[English] 日本語
 Yorodumi
Yorodumi- PDB-1kz0: Solution structure of the third helix of Antennapedia homeodomain -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1kz0 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Solution structure of the third helix of Antennapedia homeodomain | ||||||
 Components Components | Antennapedia protein | ||||||
 Keywords Keywords | DNA BINDING PROTEIN / irregular helix / turn-like parts at the beginning and at the end | ||||||
| Function / homology |  Function and homology information Function and homology informationspecification of segmental identity, antennal segment / specification of segmental identity, thorax / neuroblast development / muscle cell fate specification / lymph gland development / ventral cord development / midgut development / anterior/posterior axis specification / anterior/posterior pattern specification / regulation of neurogenesis ...specification of segmental identity, antennal segment / specification of segmental identity, thorax / neuroblast development / muscle cell fate specification / lymph gland development / ventral cord development / midgut development / anterior/posterior axis specification / anterior/posterior pattern specification / regulation of neurogenesis / heart development / sequence-specific DNA binding / DNA-binding transcription factor activity, RNA polymerase II-specific / RNA polymerase II cis-regulatory region sequence-specific DNA binding / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / DNA binding / nucleus / cytoplasm Similarity search - Function | ||||||
| Method | SOLUTION NMR / simulated annealing, molecular dynamics | ||||||
 Authors Authors | Czajlik, A. / Mesko, E. / Penke, B. / Perczel, A. | ||||||
 Citation Citation |  Journal: J.Pept.Sci. / Year: 2002 Journal: J.Pept.Sci. / Year: 2002Title: Investigation of penetratin peptides. Part 1. The environment dependent conformational properties of penetratin and two of its derivatives. Authors: Czajlik, A. / Mesko, E. / Penke, B. / Perczel, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1kz0.cif.gz 1kz0.cif.gz | 146.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1kz0.ent.gz pdb1kz0.ent.gz | 100.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1kz0.json.gz 1kz0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  1kz0_validation.pdf.gz 1kz0_validation.pdf.gz | 347 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  1kz0_full_validation.pdf.gz 1kz0_full_validation.pdf.gz | 467.8 KB | Display | |
| Data in XML |  1kz0_validation.xml.gz 1kz0_validation.xml.gz | 21.3 KB | Display | |
| Data in CIF |  1kz0_validation.cif.gz 1kz0_validation.cif.gz | 32.9 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/kz/1kz0 https://data.pdbj.org/pub/pdb/validation_reports/kz/1kz0 ftp://data.pdbj.org/pub/pdb/validation_reports/kz/1kz0 ftp://data.pdbj.org/pub/pdb/validation_reports/kz/1kz0 | HTTPS FTP |
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 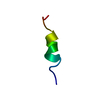
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 2253.781 Da / Num. of mol.: 1 / Fragment: C-terminal fragment of Antennapedia homeodomain / Source method: obtained synthetically Details: This sequence was synthetized in solid-phase using the Boc-chemistry. The source of this peptide is naturally found in Drosophila melanogaster (fruit fly). References: UniProt: P02833 |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR experiment | Type: 2D NOESY |
| NMR details | Text: This structure was determined using standard 2D homonuclear techniques. |
- Sample preparation
Sample preparation
| Details | Contents: 1.5mM peptide; 90% TFEd2, 10% D2O / Solvent system: 90% TFEd2, 10% D2O |
|---|---|
| Sample conditions | pH: 3 / Pressure: ambient / Temperature: 300 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker DRX / Manufacturer: Bruker / Model: DRX / Field strength: 500 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: simulated annealing, molecular dynamics / Software ordinal: 1 Details: the structures are based on a total of 553 NOE-derived restraints | ||||||||||||||||||||
| NMR representative | Selection criteria: fewest violations,lowest energy | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations,structures with the lowest energy Conformers calculated total number: 50 / Conformers submitted total number: 26 |
 Movie
Movie Controller
Controller











 PDBj
PDBj
 X-PLOR
X-PLOR