[English] 日本語
 Yorodumi
Yorodumi- PDB-1i9s: CRYSTAL STRUCTURE OF THE RNA TRIPHOSPHATASE DOMAIN OF MOUSE MRNA ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1i9s | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF THE RNA TRIPHOSPHATASE DOMAIN OF MOUSE MRNA CAPPING ENZYME | ||||||
 Components Components | MRNA CAPPING ENZYME | ||||||
 Keywords Keywords | HYDROLASE / RNA triphosphatase domain / mRNA capping enzyme | ||||||
| Function / homology |  Function and homology information Function and homology informationRNA Pol II CTD phosphorylation and interaction with CE / mRNA Capping / RNA guanylyltransferase activity / inorganic triphosphate phosphatase activity / mRNA 5'-phosphatase / mRNA 5'-triphosphate monophosphatase activity / 7-methylguanosine mRNA capping / RNA processing / phosphoprotein phosphatase activity / mRNA guanylyltransferase ...RNA Pol II CTD phosphorylation and interaction with CE / mRNA Capping / RNA guanylyltransferase activity / inorganic triphosphate phosphatase activity / mRNA 5'-phosphatase / mRNA 5'-triphosphate monophosphatase activity / 7-methylguanosine mRNA capping / RNA processing / phosphoprotein phosphatase activity / mRNA guanylyltransferase / mRNA guanylyltransferase activity / GTP binding / ATP binding / nucleus Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 1.65 Å MAD / Resolution: 1.65 Å | ||||||
 Authors Authors | Changela, A. / Ho, C.K. / Martins, A. / Shuman, S. / Mondragon, A. | ||||||
 Citation Citation |  Journal: EMBO J. / Year: 2001 Journal: EMBO J. / Year: 2001Title: Structure and mechanism of the RNA triphosphatase component of mammalian mRNA capping enzyme. Authors: Changela, A. / Ho, C.K. / Martins, A. / Shuman, S. / Mondragon, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1i9s.cif.gz 1i9s.cif.gz | 59.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1i9s.ent.gz pdb1i9s.ent.gz | 41.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1i9s.json.gz 1i9s.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/i9/1i9s https://data.pdbj.org/pub/pdb/validation_reports/i9/1i9s ftp://data.pdbj.org/pub/pdb/validation_reports/i9/1i9s ftp://data.pdbj.org/pub/pdb/validation_reports/i9/1i9s | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 24319.631 Da / Num. of mol.: 1 / Fragment: TPASE DOMAIN (RESIDUES 1-210) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Non-polymers , 5 types, 221 molecules 
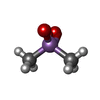







| #2: Chemical | ChemComp-SO4 / | ||
|---|---|---|---|
| #3: Chemical | ChemComp-CAC / | ||
| #4: Chemical | ChemComp-MG / | ||
| #5: Chemical | | #6: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.27 Å3/Da / Density % sol: 45.8 % | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 283 K / Method: vapor diffusion, hanging drop / pH: 6.5 Details: 15% isopropanol, 50mM sodium cacodylate (pH 6.5), 25mM magnesium chloride, 25mM ammonium sulfate , VAPOR DIFFUSION, HANGING DROP, temperature 283K | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 10 ℃ | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL9-1 / Wavelength: 0.97 Å / Beamline: BL9-1 / Wavelength: 0.97 Å |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Apr 15, 2000 |
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97 Å / Relative weight: 1 |
| Reflection | Resolution: 1.65→27 Å / Num. all: 143292 / Num. obs: 26506 / % possible obs: 99.4 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 5.4 % / Biso Wilson estimate: 16.2 Å2 / Rmerge(I) obs: 0.093 / Rsym value: 8.4 / Net I/σ(I): 5.2 |
| Reflection shell | Resolution: 1.65→1.69 Å / Redundancy: 4.9 % / Rmerge(I) obs: 0.282 / Mean I/σ(I) obs: 2.9 / Num. unique all: 1916 / Rsym value: 25.1 / % possible all: 98.6 |
| Reflection | *PLUS Num. measured all: 143292 |
| Reflection shell | *PLUS % possible obs: 98.6 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MAD / Resolution: 1.65→27 Å / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber MAD / Resolution: 1.65→27 Å / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.65→27 Å
| ||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.65→1.74 Å
| ||||||||||||||||||||||||||||||
| Software | *PLUS Name: REFMAC / Classification: refinement | ||||||||||||||||||||||||||||||
| Refinement | *PLUS σ(F): 0 / % reflection Rfree: 5 % | ||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller













 PDBj
PDBj











