[English] 日本語
 Yorodumi
Yorodumi- PDB-1hr1: Structure of an indolicidin peptide derivative with P-->A substitution -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1hr1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of an indolicidin peptide derivative with P-->A substitution | ||||||
 Components Components | INDOLICIDIN | ||||||
 Keywords Keywords | ANTIBIOTIC / ANTIMICROBIAL PROTEIN / alpha-helix / cationic antimicrobial peptide | ||||||
| Function / homology |  Function and homology information Function and homology informationAntimicrobial peptides / Neutrophil degranulation / defense response to fungus / lipopolysaccharide binding / antimicrobial humoral immune response mediated by antimicrobial peptide / killing of cells of another organism / defense response to Gram-negative bacterium / defense response to Gram-positive bacterium / innate immune response / : Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / distance geometry, simulated annealing | ||||||
 Authors Authors | Friedrich, C.L. / Rozek, A. / Patrzykat, A. / Hancock, R.E.W. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2001 Journal: J.Biol.Chem. / Year: 2001Title: Structure and mechanism of action of an indolicidin peptide derivative with improved activity against Gram-positive bacteria Authors: Friedrich, C.L. / Rozek, A. / Patrzykat, A. / Hancock, R.E.W. #1:  Journal: Biochemistry / Year: 2000 Journal: Biochemistry / Year: 2000Title: Structure of the bovine antimicrobial peptide indolicidin bound to dodecylphosphocholine and sodium dodecyl sulfate micelles. Authors: Rozek, A. / Friedrich, C.L. / Hancock, R.E.W. | ||||||
| History |
|
- Structure visualization
Structure visualization
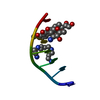
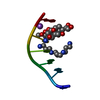
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1hr1.cif.gz 1hr1.cif.gz | 74.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1hr1.ent.gz pdb1hr1.ent.gz | 51.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1hr1.json.gz 1hr1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hr/1hr1 https://data.pdbj.org/pub/pdb/validation_reports/hr/1hr1 ftp://data.pdbj.org/pub/pdb/validation_reports/hr/1hr1 ftp://data.pdbj.org/pub/pdb/validation_reports/hr/1hr1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 1830.188 Da / Num. of mol.: 1 / Mutation: P3A,P7A,P10A / Source method: obtained synthetically Details: THE PEPTIDE WAS CHEMICALLY SYNTHESIZED. THE SEQUENCE OCCURS NATURALLY IN BOS TAURUS. THE SEQUENCE IS NATURALLY AMIDATED AT C-TERMINUS. Source: (synth.)  |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR experiment | Type: 2D NOESY |
| NMR details | Text: This structure was determined using standard 2D homonuclear techniques. |
- Sample preparation
Sample preparation
| Details | Contents: 2mM CP10A; 200mM dodecylphosphocholine / Solvent system: 10 mM phosphate buffer; 90%H2O,10%D2O |
|---|---|
| Sample conditions | Ionic strength: 200mM DPC / pH: 4 / Pressure: ambient / Temperature: 310 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker AMX / Manufacturer: Bruker / Model: AMX / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: distance geometry, simulated annealing / Software ordinal: 1 Details: The structures are based on 167 (non-redundant) NOE-derived distance restraints, 77 intraresidue and 90 inter-residue restraints. Structures were generated using DGII (MSI) and then refined using XPLOR. | ||||||||||||||||||||
| NMR representative | Selection criteria: closest to the average | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations,structures with the lowest energy Conformers calculated total number: 20 / Conformers submitted total number: 15 |
 Movie
Movie Controller
Controller










 PDBj
PDBj
 NMRPipe
NMRPipe