+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1fh1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | BACKBONE FOLD OF NODF | ||||||
 Components Components | NODULATION PROTEIN F | ||||||
 Keywords Keywords | LIPID BINDING PROTEIN / ROOT NODULATION FACTOR / PROTEIN BACKBONE FOLD | ||||||
| Function / homology | nodulation / Phosphopantetheine attachment site / Phosphopantetheine attachment site. / Phosphopantetheine attachment site / ACP-like superfamily / Carrier protein (CP) domain profile. / Phosphopantetheine binding ACP domain / Nodulation protein F Function and homology information Function and homology information | ||||||
| Biological species |  Rhizobium leguminosarum (bacteria) Rhizobium leguminosarum (bacteria) | ||||||
| Method | SOLUTION NMR / other | ||||||
 Authors Authors | Fowler, C.A. / Tian, F. / Al-Hashimi, H.M. / Prestegard, J.H. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2000 Journal: J.Mol.Biol. / Year: 2000Title: Rapid determination of protein folds using residual dipolar couplings. Authors: Fowler, C.A. / Tian, F. / Al-Hashimi, H.M. / Prestegard, J.H. #1:  Journal: FEBS Lett. / Year: 1996 Journal: FEBS Lett. / Year: 1996Title: NMR Investigations of the Structural Properties of the Nodulation Protein, NodF, from Rhizobium Leguminosarum and its Homology with Eschericia Coli Acyl Carrier Protein Authors: Ghose, R. / Geiger, O. / Prestegard, J.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1fh1.cif.gz 1fh1.cif.gz | 17.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1fh1.ent.gz pdb1fh1.ent.gz | 8.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1fh1.json.gz 1fh1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fh/1fh1 https://data.pdbj.org/pub/pdb/validation_reports/fh/1fh1 ftp://data.pdbj.org/pub/pdb/validation_reports/fh/1fh1 ftp://data.pdbj.org/pub/pdb/validation_reports/fh/1fh1 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 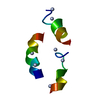
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9951.211 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Rhizobium leguminosarum (bacteria) / Plasmid: PMP2301 / Species (production host): Escherichia coli / Production host: Rhizobium leguminosarum (bacteria) / Plasmid: PMP2301 / Species (production host): Escherichia coli / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR details | Text: Assignments were made using double and triple-resonance NMR spectroscopy. Dipolar couplings were measured and used to produce the protein backbone fold. |
- Sample preparation
Sample preparation
| Details |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample conditions |
| |||||||||||||||
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: other / Software ordinal: 1 Details: secondary structural elements (3 helices) were identified. these were split into smaller fragments and individual fragments were oriented using residual dipolar coupling data and the program ...Details: secondary structural elements (3 helices) were identified. these were split into smaller fragments and individual fragments were oriented using residual dipolar coupling data and the program Orderten_SVD (Losonczi, et al., J. Magn. Res., 138, 334-342, 1999). The fragments were reassembled and then positioned spatially by translation using a limited set of NOEs to produce a backbone fold of the nodF protein. THERE ARE N-CA-C ANGLE ERRORS (AS COMPARED TO THE STANDARD DICTIONARY) AT RESIDUES 13 AND 80. RESIDUE 80 LIES SOMEWHAT OUTSIDE ALLOWED RAMACHANDRAN SPACE. THESE SITES ARE POSITIONS WHERE ORIENTED HELICAL FRAGMENTS WERE REASSEMBLED INTO COMPLETE HELICES DURING DETERMINATION OF OF THE BACKBONE FOLD AND ANY SMALLER LOCAL DISTORTIONS FROM IDEALITY ARE EXPECTED TO CONCENTRATE HERE. THE STRUCTURE PRESENTED HERE CONTAINS ONLY COORDINATES FOR BACKBONE ATOMS INVOLVED IN SECONDARY STRUCTURE. THE STRUCTURE IS THE AVERAGE OF AN ENSEMBLE WITH A HEAVY ATOM RMSD OF 2.4 ANGSTROMS. CB POSITIONS COME FROM POLYALANINE HELICES USED TO MODEL THE BACKBONE. | ||||||||||||||||||||
| NMR ensemble | Conformers submitted total number: 1 |
 Movie
Movie Controller
Controller






 PDBj
PDBj
