[English] 日本語
 Yorodumi
Yorodumi- PDB-1efm: STRUCTURE OF THE GDP DOMAIN OF EF-TU AND LOCATION OF THE AMINO AC... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1efm | ||||||
|---|---|---|---|---|---|---|---|
| Title | STRUCTURE OF THE GDP DOMAIN OF EF-TU AND LOCATION OF THE AMINO ACIDS HOMOLOGOUS TO RAS ONCOGENE PROTEINS | ||||||
 Components Components | ELONGATION FACTOR TU | ||||||
 Keywords Keywords | ELONGATION FACTOR | ||||||
| Function / homology |  Function and homology information Function and homology informationguanyl-nucleotide exchange factor complex / protein-synthesizing GTPase / guanosine tetraphosphate binding / translational elongation / translation elongation factor activity / response to antibiotic / GTPase activity / GTP binding / magnesium ion binding / RNA binding ...guanyl-nucleotide exchange factor complex / protein-synthesizing GTPase / guanosine tetraphosphate binding / translational elongation / translation elongation factor activity / response to antibiotic / GTPase activity / GTP binding / magnesium ion binding / RNA binding / plasma membrane / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.7 Å X-RAY DIFFRACTION / Resolution: 2.7 Å | ||||||
 Authors Authors | Jurnak, F. | ||||||
 Citation Citation |  Journal: Science / Year: 1985 Journal: Science / Year: 1985Title: Structure of the GDP domain of EF-Tu and location of the amino acids homologous to ras oncogene proteins. Authors: Jurnak, F. #1:  Journal: J.Biol.Chem. / Year: 1980 Journal: J.Biol.Chem. / Year: 1980Title: Biochemical and Structural Studies of the Tetragonal Crystalline Modification of the Escherichia Coli Elongation Factor TU Authors: Jurnak, F. / McPherson, A. / Wang, A.H.J. / Rich, A. #2:  Journal: J.Mol.Biol. / Year: 1977 Journal: J.Mol.Biol. / Year: 1977Title: Preliminary X-Ray Diffraction Data for Tetragonal Crystals of Trypsinized Escherichia Coli Elongation Factor Authors: Jurnak, F. / Rich, A. / Miller, D. #3:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1980 Journal: Proc.Natl.Acad.Sci.USA / Year: 1980Title: Primary Structure of Elongation Factor TU from Escherichia Coli Authors: Arai, K. / Clark, B.F.C. / Duffy, L. / Jones, M.D. / Kaziro, Y. / Laursen, R.A. / L'Italien, J. / Miller, D.L. / Nagarkatti, S. / Nakamura, S. / Nielsen, K.M. / Petersen, T.E. / Takahashi, K. / Wade, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1efm.cif.gz 1efm.cif.gz | 22.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1efm.ent.gz pdb1efm.ent.gz | 9.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1efm.json.gz 1efm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ef/1efm https://data.pdbj.org/pub/pdb/validation_reports/ef/1efm ftp://data.pdbj.org/pub/pdb/validation_reports/ef/1efm ftp://data.pdbj.org/pub/pdb/validation_reports/ef/1efm | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 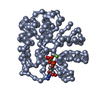
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 41651.613 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| #2: Chemical | ChemComp-MG / |
| #3: Chemical | ChemComp-GDP / |
| Compound details | TRYPSIN-MODIFIED EF-TU CONTAINS TWO MAJOR FRAGMENTS CONSISTING OF RESIDUES 1-44 AND 59-393. ...TRYPSIN-MODIFIED EF-TU CONTAINS TWO MAJOR FRAGMENTS CONSISTING |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.88 Å3/Da / Density % sol: 74.77 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS Temperature: 4,8,14,20 and 22 ℃ / pH: 7.6 / Method: vapor diffusion, hanging drop / Details: referred to J.Mol.Biol. 185.215-217 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
- Processing
Processing
| Refinement | Highest resolution: 2.7 Å | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement step | Cycle: LAST / Highest resolution: 2.7 Å
|
 Movie
Movie Controller
Controller











 PDBj
PDBj








