[English] 日本語
 Yorodumi
Yorodumi- PDB-1dcj: SOLUTION STRUCTURE OF YHHP, A NOVEL ESCHERICHIA COLI PROTEIN IMPL... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1dcj | ||||||
|---|---|---|---|---|---|---|---|
| Title | SOLUTION STRUCTURE OF YHHP, A NOVEL ESCHERICHIA COLI PROTEIN IMPLICATED IN THE CELL DIVISION | ||||||
 Components Components | YHHP PROTEIN | ||||||
 Keywords Keywords | STRUCTURAL GENOMICS / UNKNOWN FUNCTION / ALPHA-BETA SANDWICH | ||||||
| Function / homology |  Function and homology information Function and homology informationIscS-TusA complex / tRNA wobble position uridine thiolation / sulfur carrier activity / L-cysteine catabolic process / Mo-molybdopterin cofactor biosynthetic process / iron-sulfur cluster assembly / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / HYBRID DISTANCE GEOMETRY-SIMULATED ANNEALING | ||||||
 Authors Authors | Katoh, E. / Hatta, T. / Shindo, H. / Mizuno, T. / Yamazaki, T. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2000 Journal: J.Mol.Biol. / Year: 2000Title: High precision NMR structure of YhhP, a novel Escherichia coli protein implicated in cell division. Authors: Katoh, E. / Hatta, T. / Shindo, H. / Ishii, Y. / Yamada, H. / Mizuno, T. / Yamazaki, T. #1:  Journal: J.Bacteriol. / Year: 1998 Journal: J.Bacteriol. / Year: 1998Title: The yhhP Gene Encoding a Small Ubiquitous Protein is Fundamental for Normal Cell Growth of Escherichia Coli Authors: Yamashino, T. / Isomura, M. / Ueguchi, C. / Mizuno, T. #2:  Journal: To be Published Journal: To be PublishedTitle: Characterization of the yhhP Gene Whose Deficiency Results in a Non-Divided Filamentous Cell Morphology in Escherichia coli Authors: Ishii, Y. / Yamada, H. / Yamazaki, T. / Katoh, E. / Mizuno, T. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1dcj.cif.gz 1dcj.cif.gz | 491.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1dcj.ent.gz pdb1dcj.ent.gz | 412 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1dcj.json.gz 1dcj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dc/1dcj https://data.pdbj.org/pub/pdb/validation_reports/dc/1dcj ftp://data.pdbj.org/pub/pdb/validation_reports/dc/1dcj ftp://data.pdbj.org/pub/pdb/validation_reports/dc/1dcj | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 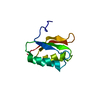
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9105.547 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||
| NMR details | Text: THE STRUCTURE WAS DETERMINED USING TRIPLE-RESONANCE NMR SPECTROSCOPY. |
- Sample preparation
Sample preparation
| Details |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample conditions |
| ||||||||||||||||||||
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker DMX / Manufacturer: Bruker / Model: DMX / Field strength: 750 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: HYBRID DISTANCE GEOMETRY-SIMULATED ANNEALING / Software ordinal: 1 Details: THE STRUCTURES ARE BASED ON A TOTAL OF 1655 RESTRAINTS, 1390 ARE NOE-DERIVED DISTANCE CONSTRAINTS, 211 DIHEDRAL ANGLE RESTRAINTS, 54 DISTANCE RESTRAINTS FROM HYDROGEN BONDS. | ||||||||||||||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 100 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller











 PDBj
PDBj NMRPipe
NMRPipe