[English] 日本語
 Yorodumi
Yorodumi- PDB-185l: SPECIFICITY OF LIGAND BINDING IN A BURIED NON-POLAR CAVITY OF T4 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 185l | ||||||
|---|---|---|---|---|---|---|---|
| Title | SPECIFICITY OF LIGAND BINDING IN A BURIED NON-POLAR CAVITY OF T4 LYSOZYME: LINKAGE OF DYNAMICS AND STRUCTURAL PLASTICITY | ||||||
 Components Components | T4 LYSOZYME | ||||||
 Keywords Keywords | HYDROLASE (O-GLYCOSYL) | ||||||
| Function / homology |  Function and homology information Function and homology informationviral release from host cell by cytolysis / peptidoglycan catabolic process / cell wall macromolecule catabolic process / lysozyme / lysozyme activity / host cell cytoplasm / defense response to bacterium Similarity search - Function | ||||||
| Biological species |  Enterobacteria phage T4 (virus) Enterobacteria phage T4 (virus) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.8 Å X-RAY DIFFRACTION / Resolution: 1.8 Å | ||||||
 Authors Authors | Morton, A. / Matthews, B.W. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1995 Journal: Biochemistry / Year: 1995Title: Specificity of ligand binding in a buried nonpolar cavity of T4 lysozyme: linkage of dynamics and structural plasticity. Authors: Morton, A. / Matthews, B.W. #1:  Journal: To be Published Journal: To be PublishedTitle: Energetic Origins of Specificity of Ligand Binding in an Interior Cavity of T4 Lysozyme Authors: Morton, A. / Baase, W.A. / Matthews, B.W. #2:  Journal: J.Mol.Biol. / Year: 1987 Journal: J.Mol.Biol. / Year: 1987Title: Structure of Bacteriophage T4 Lysozyme Refined at 1.7 Angstroms Resolution Authors: Weaver, L.H. / Matthews, B.W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  185l.cif.gz 185l.cif.gz | 47.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb185l.ent.gz pdb185l.ent.gz | 33.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  185l.json.gz 185l.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/85/185l https://data.pdbj.org/pub/pdb/validation_reports/85/185l ftp://data.pdbj.org/pub/pdb/validation_reports/85/185l ftp://data.pdbj.org/pub/pdb/validation_reports/85/185l | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  181lC  182lC  183lC  184lC  186lC  187lC  188lC  1nhbC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 18586.283 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Enterobacteria phage T4 (virus) / Genus: T4-like viruses / Species: Enterobacteria phage T4 sensu lato / Plasmid: M13 / References: UniProt: P00720, lysozyme Enterobacteria phage T4 (virus) / Genus: T4-like viruses / Species: Enterobacteria phage T4 sensu lato / Plasmid: M13 / References: UniProt: P00720, lysozyme | ||||||
|---|---|---|---|---|---|---|---|
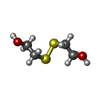
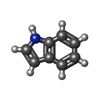
| #2: Chemical | | #3: Chemical | ChemComp-HED / | #4: Chemical | ChemComp-IND / | #5: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.79 Å3/Da / Density % sol: 55.95 % | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop / PH range low: 7.1 / PH range high: 6.3 | ||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| Reflection | *PLUS Highest resolution: 1.8 Å / Lowest resolution: 20 Å / Num. obs: 41721 / % possible obs: 79 % / Observed criterion σ(I): 0 |
- Processing
Processing
| Software | Name: TNT / Classification: refinement | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.8→20 Å / σ(F): 0 /
| ||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→20 Å
| ||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj