登録情報 データベース : EMDB / ID : EMD-9101タイトル The 20S supercomplex engaging the SNAP-25 N-terminus (class 2) The unsharpened map. 複合体 : 20S supercomplex consisting of soluble neuronal SNARE complex, alpha-SNAP, and N-ethylmaleimide sensitive factor (NSF)複合体 : N-ethylmaleimide sensitive factorタンパク質・ペプチド : Vesicle-fusing ATPase複合体 : Synaptosomal-associated protein 25タンパク質・ペプチド : Synaptosomal-associated protein 25複合体 : Syntaxin-1A複合体 : Vesicle-associated membrane protein 2タンパク質・ペプチド : Vesicle-associated membrane protein 2複合体 : Alpha-soluble NSF attachment proteinタンパク質・ペプチド : Alpha-soluble NSF attachment proteinリガンド : ADENOSINE-5'-TRIPHOSPHATEリガンド : ADENOSINE-5'-DIPHOSPHATE / / / / / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
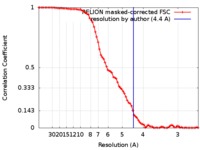
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Cricetulus griseus (モンゴルキヌゲネズミ) / Rattus norvegicus (ドブネズミ)手法 / / 解像度 : 4.4 Å White KI / Zhao M 資金援助 Organization Grant number 国 National Institutes of Health/National Human Genome Research Institute (NIH/NHGRI) R37MH63105
ジャーナル : Elife / 年 : 2018タイトル : Structural principles of SNARE complex recognition by the AAA+ protein NSF.著者 : K Ian White / Minglei Zhao / Ucheor B Choi / Richard A Pfuetzner / Axel T Brunger / 要旨 : The recycling of SNARE proteins following complex formation and membrane fusion is an essential process in eukaryotic trafficking. A highly conserved AAA+ protein, NSF (-ethylmaleimide sensitive ... The recycling of SNARE proteins following complex formation and membrane fusion is an essential process in eukaryotic trafficking. A highly conserved AAA+ protein, NSF (-ethylmaleimide sensitive factor) and an adaptor protein, SNAP (soluble NSF attachment protein), disassemble the SNARE complex. We report electron-cryomicroscopy structures of the complex of NSF, αSNAP, and the full-length soluble neuronal SNARE complex (composed of syntaxin-1A, synaptobrevin-2, SNAP-25A) in the presence of ATP under non-hydrolyzing conditions at ~3.9 Å resolution. These structures reveal electrostatic interactions by which two αSNAP molecules interface with a specific surface of the SNARE complex. This interaction positions the SNAREs such that the 15 N-terminal residues of SNAP-25A are loaded into the D1 ring pore of NSF via a spiral pattern of interactions between a conserved tyrosine NSF residue and SNAP-25A backbone atoms. This loading process likely precedes ATP hydrolysis. Subsequent ATP hydrolysis then drives complete disassembly. 履歴 登録 2018年9月4日 - ヘッダ(付随情報) 公開 2018年9月19日 - マップ公開 2018年9月19日 - 更新 2024年3月13日 - 現状 2024年3月13日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報

 データ登録者
データ登録者 米国, 1件
米国, 1件  引用
引用 ジャーナル: Elife / 年: 2018
ジャーナル: Elife / 年: 2018
 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_9101.map.gz
emd_9101.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-9101-v30.xml
emd-9101-v30.xml emd-9101.xml
emd-9101.xml EMDBヘッダ
EMDBヘッダ emd_9101_fsc.xml
emd_9101_fsc.xml FSCデータファイル
FSCデータファイル emd_9101.png
emd_9101.png emd-9101.cif.gz
emd-9101.cif.gz emd_9101_additional.map.gz
emd_9101_additional.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-9101
http://ftp.pdbj.org/pub/emdb/structures/EMD-9101 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9101
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9101 emd_9101_validation.pdf.gz
emd_9101_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_9101_full_validation.pdf.gz
emd_9101_full_validation.pdf.gz emd_9101_validation.xml.gz
emd_9101_validation.xml.gz emd_9101_validation.cif.gz
emd_9101_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9101
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9101 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9101
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9101 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
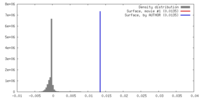
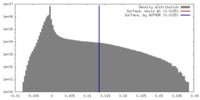
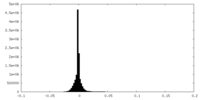
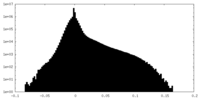
マップ ダウンロード / ファイル: emd_9101.map.gz / 形式: CCP4 / 大きさ: 46.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_9101.map.gz / 形式: CCP4 / 大きさ: 46.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー




























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)