[English] 日本語
 Yorodumi
Yorodumi- EMDB-8009: Combined Body and arm regions of the yeast spliceosomal U4/U6.U5 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8009 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Combined Body and arm regions of the yeast spliceosomal U4/U6.U5 tri-snRNP by masked classification and refinement-Class 2 | |||||||||
 Map data Map data | Combined body and arm regions of the yeast spliceosomal U4/U6.U5 tri-snRNP from masked classification and refinement-class2 | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.5 Å | |||||||||
 Authors Authors | Nguyen THD / Galej WP / Bai X-C / Oubridge C / Newman AJ / Scheres SHW / Nagai K | |||||||||
 Citation Citation |  Journal: Nature / Year: 2016 Journal: Nature / Year: 2016Title: Cryo-EM structure of the yeast U4/U6.U5 tri-snRNP at 3.7 Å resolution. Authors: Thi Hoang Duong Nguyen / Wojciech P Galej / Xiao-Chen Bai / Chris Oubridge / Andrew J Newman / Sjors H W Scheres / Kiyoshi Nagai /  Abstract: U4/U6.U5 tri-snRNP represents a substantial part of the spliceosome before activation. A cryo-electron microscopy structure of Saccharomyces cerevisiae U4/U6.U5 tri-snRNP at 3.7 Å resolution led ...U4/U6.U5 tri-snRNP represents a substantial part of the spliceosome before activation. A cryo-electron microscopy structure of Saccharomyces cerevisiae U4/U6.U5 tri-snRNP at 3.7 Å resolution led to an essentially complete atomic model comprising 30 proteins plus U4/U6 and U5 small nuclear RNAs (snRNAs). The structure reveals striking interweaving interactions of the protein and RNA components, including extended polypeptides penetrating into subunit interfaces. The invariant ACAGAGA sequence of U6 snRNA, which base-pairs with the 5'-splice site during catalytic activation, forms a hairpin stabilized by Dib1 and Prp8 while the adjacent nucleotides interact with the exon binding loop 1 of U5 snRNA. Snu114 harbours GTP, but its putative catalytic histidine is held away from the γ-phosphate by hydrogen bonding to a tyrosine in the amino-terminal domain of Prp8. Mutation of this histidine to alanine has no detectable effect on yeast growth. The structure provides important new insights into the spliceosome activation process leading to the formation of the catalytic centre. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8009.map.gz emd_8009.map.gz | 195.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8009-v30.xml emd-8009-v30.xml emd-8009.xml emd-8009.xml | 46 KB 46 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_8009_fsc.xml emd_8009_fsc.xml | 13.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_8009.png emd_8009.png | 72.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8009 http://ftp.pdbj.org/pub/emdb/structures/EMD-8009 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8009 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8009 | HTTPS FTP |
-Related structure data
| Related structure data |  8006C  8007C  8008C  8010C  8011C  8012C  8013C  8014C  5gamC  5ganC  5gaoC  5gapC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_8009.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8009.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Combined body and arm regions of the yeast spliceosomal U4/U6.U5 tri-snRNP from masked classification and refinement-class2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.43 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
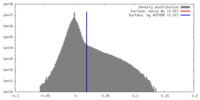
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : tri-snRNP complex with 30 proteins and 3 snRNA
+Supramolecule #1: tri-snRNP complex with 30 proteins and 3 snRNA
+Macromolecule #1: U4 snRNA
+Macromolecule #2: U6 snRNA
+Macromolecule #21: U5 snRNA
+Macromolecule #3: Prp8
+Macromolecule #4: Prp4
+Macromolecule #5: Prp6
+Macromolecule #6: Dib1
+Macromolecule #7: Prp31
+Macromolecule #8: Prp3
+Macromolecule #9: Brr2
+Macromolecule #10: unknown proteins
+Macromolecule #11: SmB
+Macromolecule #12: U4 SmD1
+Macromolecule #13: SmD2
+Macromolecule #14: SmD3
+Macromolecule #15: SmE
+Macromolecule #16: SmF
+Macromolecule #17: SmG
+Macromolecule #18: Snu66
+Macromolecule #19: U5 SmE
+Macromolecule #20: U5 SmD1
+Macromolecule #22: Snu13
+Macromolecule #23: LSm2
+Macromolecule #24: LSm3
+Macromolecule #25: LSm4
+Macromolecule #26: LSm5
+Macromolecule #27: LSm6
+Macromolecule #28: LSm7
+Macromolecule #29: LSm8
+Macromolecule #30: Snu114
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL |
|---|---|
| Buffer | pH: 7.9 / Component - Concentration: 1.0 mM / Component - Name: DTT |
| Grid | Model: Quantifoil R 1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 6.0 nm / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: OTHER |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-20 / Average exposure time: 16.0 sec. / Average electron dose: 38.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 35714 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)