[English] 日本語
 Yorodumi
Yorodumi- EMDB-7807: Subtomogram average (735 axonemal repeats) of isolated Rib72B kno... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7807 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Subtomogram average (735 axonemal repeats) of isolated Rib72B knock out Tetrahymena cilia Rescued with Rib72B-GFP, showing the lumen of the ciliary doublet microtubule (DMT) | |||||||||
 Map data Map data | Rib72BKO Rescued with Rib72B-GFP | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | subtomogram averaging / cryo EM / Resolution: 40.1 Å | |||||||||
 Authors Authors | Stoddard D / Zhao Y / Bayless B / Gui L / Louka P / Dave D / Surwayanshi S / Tomasi R / Dupuis-Williams P / Baroud C ...Stoddard D / Zhao Y / Bayless B / Gui L / Louka P / Dave D / Surwayanshi S / Tomasi R / Dupuis-Williams P / Baroud C / Gaertig J / Winey M / Nicastro D | |||||||||
 Citation Citation |  Journal: Mol Biol Cell / Year: 2018 Journal: Mol Biol Cell / Year: 2018Title: Tetrahymena RIB72A and RIB72B are microtubule inner proteins in the ciliary doublet microtubules. Authors: Daniel Stoddard / Ying Zhao / Brian A Bayless / Long Gui / Panagiota Louka / Drashti Dave / Swati Suryawanshi / Raphaël F-X Tomasi / Pascale Dupuis-Williams / Charles N Baroud / Jacek ...Authors: Daniel Stoddard / Ying Zhao / Brian A Bayless / Long Gui / Panagiota Louka / Drashti Dave / Swati Suryawanshi / Raphaël F-X Tomasi / Pascale Dupuis-Williams / Charles N Baroud / Jacek Gaertig / Mark Winey / Daniela Nicastro /   Abstract: Doublet and triplet microtubules are essential and highly stable core structures of centrioles, basal bodies, cilia, and flagella. In contrast to dynamic cytoplasmic micro-tubules, their luminal ...Doublet and triplet microtubules are essential and highly stable core structures of centrioles, basal bodies, cilia, and flagella. In contrast to dynamic cytoplasmic micro-tubules, their luminal surface is coated with regularly arranged microtubule inner proteins (MIPs). However, the protein composition and biological function(s) of MIPs remain poorly understood. Using genetic, biochemical, and imaging techniques, we identified Tetrahymena RIB72A and RIB72B proteins as ciliary MIPs. Fluorescence imaging of tagged RIB72A and RIB72B showed that both proteins colocalize to Tetrahymena cilia and basal bodies but assemble independently. Cryoelectron tomography of RIB72A and/or RIB72B knockout strains revealed major structural defects in the ciliary A-tubule involving MIP1, MIP4, and MIP6 structures. The defects of individual mutants were complementary in the double mutant. All mutants had reduced swimming speed and ciliary beat frequencies, and high-speed video imaging revealed abnormal highly curved cilia during power stroke. Our results show that RIB72A and RIB72B are crucial for the structural assembly of ciliary A-tubule MIPs and are important for proper ciliary motility. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7807.map.gz emd_7807.map.gz | 3.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7807-v30.xml emd-7807-v30.xml emd-7807.xml emd-7807.xml | 12.8 KB 12.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7807.png emd_7807.png | 75.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7807 http://ftp.pdbj.org/pub/emdb/structures/EMD-7807 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7807 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7807 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_7807.map.gz / Format: CCP4 / Size: 3.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7807.map.gz / Format: CCP4 / Size: 3.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Rib72BKO Rescued with Rib72B-GFP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 10.77 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
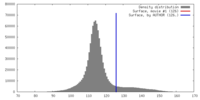
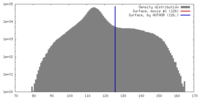
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Subtomogram average (735 axonemal repeats) of isolated Rib72B kno...
| Entire | Name: Subtomogram average (735 axonemal repeats) of isolated Rib72B knock out Tetrahymena cilia Rescued with Rib72B-GFP, showing the lumen of the ciliary doublet microtubule (DMT) |
|---|---|
| Components |
|
-Supramolecule #1: Subtomogram average (735 axonemal repeats) of isolated Rib72B kno...
| Supramolecule | Name: Subtomogram average (735 axonemal repeats) of isolated Rib72B knock out Tetrahymena cilia Rescued with Rib72B-GFP, showing the lumen of the ciliary doublet microtubule (DMT) type: organelle_or_cellular_component / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | tissue |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Component:
Details: Solutions were freshly made before use | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 101.325 kPa | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber temperature: 100 K / Instrument: HOMEMADE PLUNGER Details: back-side blotting for 1.5-2.5 seconds before plunging. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F30 |
|---|---|
| Specialist optics | Energy filter - Name: GIF 2000 / Energy filter - Lower energy threshold: 0 eV / Energy filter - Upper energy threshold: 20 eV |
| Image recording | Film or detector model: GATAN ULTRASCAN 1000 (2k x 2k) / Average electron dose: 1.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 8.0 µm / Calibrated magnification: 13500 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus min: 6.0 µm |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 40.1 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name:  IMOD / Number subtomograms used: 735 IMOD / Number subtomograms used: 735 |
|---|---|
| Extraction | Number tomograms: 5 / Number images used: 735 / Software - Name:  MATLAB MATLAB |
| Final angle assignment | Type: ANGULAR RECONSTITUTION / Software - Name: PEET |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)