[English] 日本語
 Yorodumi
Yorodumi- EMDB-7077: Model for compact volume of truncated monomeric Cytohesin-3 (Grp1... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7077 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Model for compact volume of truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A 6GS Arf6 Q67L fusion protein | |||||||||
 Map data Map data | Compact volume for truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A 6GS Arf6 Q67L fusion protein | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Guanine nucleotide exchange factor / Arf GTPase / Fusion protein / Inositol 1 / 3 / 4 / 5-tetrakisphosphate / LIPID BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationerythrocyte apoptotic process / maintenance of postsynaptic density structure / Intra-Golgi traffic / protein localization to cleavage furrow / positive regulation of mitotic cytokinetic process / establishment of epithelial cell polarity / Golgi vesicle transport / regulation of dendritic spine development / negative regulation of protein localization to cell surface / protein localization to endosome ...erythrocyte apoptotic process / maintenance of postsynaptic density structure / Intra-Golgi traffic / protein localization to cleavage furrow / positive regulation of mitotic cytokinetic process / establishment of epithelial cell polarity / Golgi vesicle transport / regulation of dendritic spine development / negative regulation of protein localization to cell surface / protein localization to endosome / regulation of Rac protein signal transduction / negative regulation of dendrite development / ruffle assembly / regulation of filopodium assembly / positive regulation of keratinocyte migration / positive regulation of focal adhesion disassembly / endocytic recycling / MET receptor recycling / thioesterase binding / regulation of ARF protein signal transduction / Flemming body / filopodium membrane / TBC/RABGAPs / protein localization to cell surface / cortical actin cytoskeleton organization / hepatocyte apoptotic process / phosphatidylinositol-3,4,5-trisphosphate binding / positive regulation of actin filament polymerization / cleavage furrow / bicellular tight junction / endocytic vesicle / regulation of presynapse assembly / synaptic vesicle endocytosis / vesicle-mediated transport / ruffle / signaling adaptor activity / guanyl-nucleotide exchange factor activity / positive regulation of cell adhesion / positive regulation of protein secretion / small monomeric GTPase / protein localization to plasma membrane / positive regulation of protein localization to plasma membrane / adherens junction / liver development / negative regulation of receptor-mediated endocytosis / cellular response to nerve growth factor stimulus / intracellular protein transport / positive regulation of neuron projection development / recycling endosome membrane / GDP binding / nervous system development / Clathrin-mediated endocytosis / presynapse / G protein activity / midbody / early endosome membrane / cell cortex / cell differentiation / cell adhesion / endosome / postsynapse / cell division / focal adhesion / GTPase activity / GTP binding / glutamatergic synapse / Golgi apparatus / extracellular exosome / nucleoplasm / membrane / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 35.0 Å | |||||||||
 Authors Authors | Das S / Malaby AW | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Structure / Year: 2018 Journal: Structure / Year: 2018Title: Structural Dynamics Control Allosteric Activation of Cytohesin Family Arf GTPase Exchange Factors. Authors: Andrew W Malaby / Sanchaita Das / Srinivas Chakravarthy / Thomas C Irving / Osman Bilsel / David G Lambright /  Abstract: Membrane dynamic processes including vesicle biogenesis depend on Arf guanosine triphosphatase (GTPase) activation by guanine nucleotide exchange factors (GEFs) containing a catalytic Sec7 domain and ...Membrane dynamic processes including vesicle biogenesis depend on Arf guanosine triphosphatase (GTPase) activation by guanine nucleotide exchange factors (GEFs) containing a catalytic Sec7 domain and a membrane-targeting module such as a pleckstrin homology (PH) domain. The catalytic output of cytohesin family Arf GEFs is controlled by autoinhibitory interactions that impede accessibility of the exchange site in the Sec7 domain. These restraints can be relieved through activator Arf-GTP binding to an allosteric site comprising the PH domain and proximal autoinhibitory elements (Sec7-PH linker and C-terminal helix). Small-angle X-ray scattering and negative-stain electron microscopy were used to investigate the structural organization and conformational dynamics of cytohesin-3 (Grp1) in autoinhibited and active states. The results support a model in which hinge dynamics in the autoinhibited state expose the activator site for Arf-GTP binding, while subsequent C-terminal helix unlatching and repositioning unleash conformational entropy in the Sec7-PH linker to drive exposure of the exchange site. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7077.map.gz emd_7077.map.gz | 310.1 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7077-v30.xml emd-7077-v30.xml emd-7077.xml emd-7077.xml | 13.8 KB 13.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7077.png emd_7077.png | 20.7 KB | ||
| Filedesc metadata |  emd-7077.cif.gz emd-7077.cif.gz | 5.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7077 http://ftp.pdbj.org/pub/emdb/structures/EMD-7077 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7077 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7077 | HTTPS FTP |
-Related structure data
| Related structure data |  6bbpMC  7078C  6bbqC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7077.map.gz / Format: CCP4 / Size: 2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7077.map.gz / Format: CCP4 / Size: 2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Compact volume for truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A 6GS Arf6 Q67L fusion protein | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A ...
| Entire | Name: Truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A 6GS Arf6 Q67L fusion protein complex with GTP, Mg and Inositol 1,3,4,5 tetrakisphosphate |
|---|---|
| Components |
|
-Supramolecule #1: Truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A ...
| Supramolecule | Name: Truncated monomeric Cytohesin-3 (Grp1; amino acids 63-399) E161A 6GS Arf6 Q67L fusion protein complex with GTP, Mg and Inositol 1,3,4,5 tetrakisphosphate type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Cytohesin-3,ADP-ribosylation factor 6
| Macromolecule | Name: Cytohesin-3,ADP-ribosylation factor 6 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 60.292777 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGHHHHHHGS TTQRNKQIAM GRKKFNMDPK KGIQFLIEND LLQSSPEDVA QFLYKGEGLN KTVIGDYLGE RDDFNIKVLQ AFVELHEFA DLNLVQALRQ FLWSFRLPGE AQKIDRMMEA FASRYCLCNP GVFQSTDTCY VLSFAIIMLN TSLHNHNVRD K PTAERFIT ...String: MGHHHHHHGS TTQRNKQIAM GRKKFNMDPK KGIQFLIEND LLQSSPEDVA QFLYKGEGLN KTVIGDYLGE RDDFNIKVLQ AFVELHEFA DLNLVQALRQ FLWSFRLPGE AQKIDRMMEA FASRYCLCNP GVFQSTDTCY VLSFAIIMLN TSLHNHNVRD K PTAERFIT MNRGINEGGD LPEELLRNLY ESIKNEPFKI PEDDGNDLTH TFFNPDREGW LLKLGGRVKT WKRRWFILTD NC LYYFEYT TDKEPRGIIP LENLSIREVE DPRKPNCFEL YNPSHKGQVI KACKTEADGR VVEGNHVVYR ISAPSPEEKE EWM KSIKAS ISRDPFYDML ATRKRRIANK KGKVLSKIFG NKEMRILMLG LDAAGKTTIL YKLKLGQSVT TIPTVGFNVE TVTY KNVKF NVWDVGGLDK IRPLWRHYYT GTQGLIFVVD CADRDRIDEA RQELHRIIND REMRDAIILI FANKQDLPDA MKPHE IQEK LGLTRIRDRN WYVQPSCATS GDGLYEGLTW LTSNYN UniProtKB: Cytohesin-3, ADP-ribosylation factor 6 |
-Macromolecule #2: GUANOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: GUANOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 1 / Formula: GTP |
|---|---|
| Molecular weight | Theoretical: 523.18 Da |
| Chemical component information |  ChemComp-GTP: |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #4: INOSITOL-(1,3,4,5)-TETRAKISPHOSPHATE
| Macromolecule | Name: INOSITOL-(1,3,4,5)-TETRAKISPHOSPHATE / type: ligand / ID: 4 / Number of copies: 1 / Formula: 4IP |
|---|---|
| Molecular weight | Theoretical: 500.075 Da |
| Chemical component information | 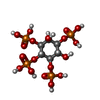 ChemComp-4IP: |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Details: 20 mM Tris, pH 8.0, 150 mM NaCl, 2 mM MgCl2, 0.1% 2-mercaptoethanol, and 0.001 mM IP4 |
|---|---|
| Staining | Type: NEGATIVE / Material: Uranyl Formate / Details: Stained with 0.75% (w/v) uranyl formate |
| Grid | Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. / Pretreatment - Atmosphere: AIR |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 12 |
|---|---|
| Details | Gatan Erlang Shen 785 camera used for collecting images |
| Image recording | Film or detector model: OTHER / Number grids imaged: 1 / Number real images: 369 / Average electron dose: 20.0 e/Å2 Details: Gatan Erlang Shen 785 camera used for collecting images |
| Electron beam | Acceleration voltage: 120 kV / Electron source: LAB6 |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 60000 |
 Movie
Movie Controller
Controller


 UCSF Chimera
UCSF Chimera




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)