+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7042 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the mechanosensitive channel Piezo1 | |||||||||
 Map data Map data | Mechanosensitive channel Piezo1, focus-refined map in C1, sharpened with a B-factor of -200A2, low-passed to 3.8A | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | triskelion / transmembrane / ion channel / MEMBRANE PROTEIN / TRANSPORT PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationmechanosensitive monoatomic cation channel activity / cuticular plate / positive regulation of integrin activation / detection of mechanical stimulus / positive regulation of cell-cell adhesion mediated by integrin / mechanosensitive monoatomic ion channel activity / stereocilium / positive regulation of myotube differentiation / lamellipodium membrane / monoatomic cation transport ...mechanosensitive monoatomic cation channel activity / cuticular plate / positive regulation of integrin activation / detection of mechanical stimulus / positive regulation of cell-cell adhesion mediated by integrin / mechanosensitive monoatomic ion channel activity / stereocilium / positive regulation of myotube differentiation / lamellipodium membrane / monoatomic cation transport / monoatomic cation channel activity / endoplasmic reticulum-Golgi intermediate compartment membrane / cellular response to mechanical stimulus / regulation of membrane potential / endoplasmic reticulum membrane / endoplasmic reticulum / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Guo YR / MacKinnon R | |||||||||
 Citation Citation |  Journal: Elife / Year: 2017 Journal: Elife / Year: 2017Title: Structure-based membrane dome mechanism for Piezo mechanosensitivity. Authors: Yusong R Guo / Roderick MacKinnon /  Abstract: Mechanosensitive ion channels convert external mechanical stimuli into electrochemical signals for critical processes including touch sensation, balance, and cardiovascular regulation. The best ...Mechanosensitive ion channels convert external mechanical stimuli into electrochemical signals for critical processes including touch sensation, balance, and cardiovascular regulation. The best understood mechanosensitive channel, MscL, opens a wide pore, which accounts for mechanosensitive gating due to in-plane area expansion. Eukaryotic Piezo channels have a narrow pore and therefore must capture mechanical forces to control gating in another way. We present a cryo-EM structure of mouse Piezo1 in a closed conformation at 3.7Å-resolution. The channel is a triskelion with arms consisting of repeated arrays of 4-TM structural units surrounding a pore. Its shape deforms the membrane locally into a dome. We present a hypothesis in which the membrane deformation changes upon channel opening. Quantitatively, membrane tension will alter gating energetics in proportion to the change in projected area under the dome. This mechanism can account for highly sensitive mechanical gating in the setting of a narrow, cation-selective pore. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7042.map.gz emd_7042.map.gz | 228.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7042-v30.xml emd-7042-v30.xml emd-7042.xml emd-7042.xml | 18 KB 18 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_7042_fsc_1.xml emd_7042_fsc_1.xml emd_7042_fsc_2.xml emd_7042_fsc_2.xml | 14 KB 16.4 KB | Display Display |  FSC data file FSC data file |
| Images |  emd_7042_1.png emd_7042_1.png emd_7042_2.png emd_7042_2.png | 63.4 KB 58.1 KB | ||
| Filedesc metadata |  emd-7042.cif.gz emd-7042.cif.gz | 7.4 KB | ||
| Others |  emd_7042_additional.map.gz emd_7042_additional.map.gz | 228.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7042 http://ftp.pdbj.org/pub/emdb/structures/EMD-7042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7042 | HTTPS FTP |
-Related structure data
| Related structure data |  6b3rMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7042.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7042.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Mechanosensitive channel Piezo1, focus-refined map in C1, sharpened with a B-factor of -200A2, low-passed to 3.8A | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
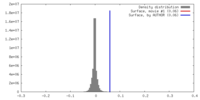
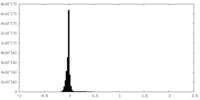
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Mechanosensitive channel Piezo1, full map refined in C3
| File | emd_7042_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Mechanosensitive channel Piezo1, full map refined in C3 | ||||||||||||
| Projections & Slices |
| ||||||||||||
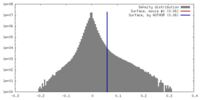
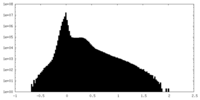
| Density Histograms |
- Sample components
Sample components
-Entire : mouse Piezo1 channel
| Entire | Name: mouse Piezo1 channel |
|---|---|
| Components |
|
-Supramolecule #1: mouse Piezo1 channel
| Supramolecule | Name: mouse Piezo1 channel / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 900 KDa |
-Macromolecule #1: Piezo-type mechanosensitive ion channel component 1
| Macromolecule | Name: Piezo-type mechanosensitive ion channel component 1 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 292.320656 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MEPHVLGAGL YWLLLPCTLL AASLLRFNAL SLVYLLFLLL LPWLPGPSRH SIPGHTGRLL RALLCLSLLF LVAHLAFQIC LHTVPHLDQ FLGQNGSLWV KVSQHIGVTR LDLKDIFNTT RLVAPDLGVL LASSLCLGLC GRLTRKAGQS RRTQELQDDD D DDDDDDED ...String: MEPHVLGAGL YWLLLPCTLL AASLLRFNAL SLVYLLFLLL LPWLPGPSRH SIPGHTGRLL RALLCLSLLF LVAHLAFQIC LHTVPHLDQ FLGQNGSLWV KVSQHIGVTR LDLKDIFNTT RLVAPDLGVL LASSLCLGLC GRLTRKAGQS RRTQELQDDD D DDDDDDED IDAAPAVGLK GAPALATKRR LWLASRFRVT AHWLLMTSGR TLVIVLLALA GIAHPSAFSS IYLVVFLAIC TW WSCHFPL SPLGFNTLCV MVSCFGAGHL ICLYCYQTPF IQDMLPPGNI WARLFGLKNF VDLPNYSSPN ALVLNTKHAW PIY VSPGIL LLLYYTATSL LKLHKSCPSE LRKETPREDE EHELELDHLE PEPQARDATQ GEMPMTTEPD LDNCTVHVLT SQSP VRQRP VRPRLAELKE MSPLHGLGHL IMDQSYVCAL IAMMVWSIMY HSWLTFVLLL WACLIWTVRS RHQLAMLCSP CILLY GLTL CCLRYVWAME LPELPTTLGP VSLHQLGLEH TRYPCLDLGA MLLYLLTFWL LLRQFVKEKL LKKQKVPAAL LEVTVA DTE PTQTQTLLRS LGELVTGIYV KYWIYVCAGM FIVVSFAGRL VVYKIVYMFL FLLCLTLFQV YYTLWRKLLR VFWWLVV AY TMLVLIAVYT FQFQDFPTYW RNLTGFTDEQ LGDLGLEQFS VSELFSSILI PGFFLLACIL QLHYFHRPFM QLTDLEHV P PPGTRHPRWA HRQDAVSEAP LLEHQEEEEV FREDGQSMDG PHQATQVPEG TASKWGLVAD RLLDLAASFS AVLTRIQVF VRRLLELHVF KLVALYTVWV ALKEVSVMNL LLVVLWAFAL PYPRFRPMAS CLSTVWTCII IVCKMLYQLK IVNPHEYSSN CTEPFPNNT NLQPLEINQS LLYRGPVDPA NWFGVRKGYP NLGYIQNHLQ ILLLLVFEAV VYRRQEHYRR QHQQAPLPAQ A VCADGTRQ RLDQDLLSCL KYFINFFFYK FGLEICFLMA VNVIGQRMNF MVILHGCWLV AILTRRRREA IARLWPNYCL FL TLFLLYQ YLLCLGMPPA LCIDYPWRWS KAIPMNSALI KWLYLPDFFR APNSTNLISD FLLLLCASQQ WQVFSAERTE EWQ RMAGIN TDHLEPLRGE PNPIPNFIHC RSYLDMLKVA VFRYLFWLVL VVVFVAGATR ISIFGLGYLL ACFYLLLFGT TLLQ KDTRA QLVLWDCLIL YNVTVIISKN MLSLLSCVFV EQMQSNFCWV IQLFSLVCTV KGYYDPKEMM TRDRDCLLPV EEAGI IWDS ICFFFLLLQR RIFLSHYFLH VSADLKATAL QASRGFALYN AANLKSINFH RQIEEKSLAQ LKRQMKRIRA KQEKYR QSQ ASRGQLQSKD PQDPSQEPGP DSPGGSSPPR RQWWRPWLDH ATVIHSGDYF LFESDSEEEE EALPEDPRPA AQSAFQM AY QAWVTNAQTV LRQRRERARQ ERAEQLASGG DLNPDVEPVD VPEDEMAGRS HMMQRVLSTM QFLWVLGQAT VDGLTRWL R AFTKHHRTMS DVLCAERYLL TQELLRVGEV RRGVLDQLYV GEDEATLSGP VETRDGPSTA SSGLGAEEPL SSMTDDTSS PLSTGYNTRS GSEEIVTDAG DLQAGTSLHG SQELLANART RMRTASELLL DRRLHIPELE EAERFEAQQG RTLRLLRAGY QCVAAHSEL LCYFIIILNH MVTASAASLV LPVLVFLWAM LTIPRPSKRF WMTAIVFTEV MVVTKYLFQF GFFPWNSYVV L RRYENKPY FPPRILGLEK TDSYIKYDLV QLMALFFHRS QLLCYGLWDH EEDRYPKDHC RSSVKDREAK EEPEAKLESQ SE TGTGHPK EPVLAGTPRD HIQGKGSIRS KDVIQDPPED LKPRHTRHIS IRFRRRKETP GPKGTAVMET EHEEGEGKET TER KRPRHT QEKSKFRERM KAAGRRLQSF CVSLAQSFYQ PLQRFFHDIL HTKYRAATDV YALMFLADIV DIIIIIFGFW AFGK HSAAT DIASSLSDDQ VPQAFLFMLL VQFGTMVIDR ALYLRKTVLG KLAFQVVLVV AIHIWMFFIL PAVTERMFSQ NAVAQ LWYF VKCIYFALSA YQIRCGYPTR ILGNFLTKKY NHLNLFLFQG FRLVPFLVEL RAVMDWVWTD TTLSLSNWMC VEDIYA NIF IIKCSRETEK KYPQPKGQKK KKIVKYGMGG LIILFLIAII WFPLLFMSLI RSVVGVVNQP IDVTVTLKLG GYEPLFT MS AQQPSIVPFT PQAYEELSQQ FDPYPLAMQF ISQYSPEDIV TAQIEGSSGA LWRISPPSRA QMKQELYNGT ADITLRFT W NFQRDLAKGG TVEYTNEKHT LELAPNSTAR RQLAQLLEGR PDQSVVIPHL FPKYIRAPNG PEANPVKQLQ PDEEEDYLG VRIQLRREQV GTGASGEQAG TKASDFLEWW VIELQDCKAD CNLLPMVIFS DKVSPPSLGF LAGYGIVGLY VSIVLVVGKF VRGFFSEIS HSIMFEELPC VDRILKLCQD IFLVRETREL ELEEELYAKL IFLYRSPETM IKWTRERE UniProtKB: Piezo-type mechanosensitive ion channel component 1 |
-Macromolecule #2: Piezo-type mechanosensitive ion channel component 1, unknown fragment
| Macromolecule | Name: Piezo-type mechanosensitive ion channel component 1, unknown fragment type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.379692 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 15 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 400 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 10 sec. / Pretreatment - Atmosphere: OTHER | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK IV / Details: blot for 1 second with -1 force before plunging. | ||||||||||||
| Details | This sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-50 / Number grids imaged: 1 / Number real images: 3414 / Average exposure time: 10.0 sec. / Average electron dose: 47.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)