+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of CpcL-PBS2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Phycobilisomes / PHOTOSYNTHESIS | |||||||||
| Function / homology |  Function and homology information Function and homology informationphycobilisome / plasma membrane-derived thylakoid membrane / photosynthesis Similarity search - Function | |||||||||
| Biological species |  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.58 Å | |||||||||
 Authors Authors | Mao ZY / Li ZH / Han GY | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2026 Journal: Proc Natl Acad Sci U S A / Year: 2026Title: Structural insight of a photosystem I-CpcL-phycobilisome supercomplex from a cyanobacterium sp. PCC 7120. Authors: Zhiyuan Mao / Zhenhua Li / Xingyue Li / Liangliang Shen / Tingyun Kuang / Wenda Wang / Jian-Ren Shen / Guangye Han /   Abstract: Phycobilisomes (PBSs) are supramolecular pigment-protein complexes composed of phycobiliproteins and linker proteins, serving as the major light-harvesting complexes that capture and transfer light ...Phycobilisomes (PBSs) are supramolecular pigment-protein complexes composed of phycobiliproteins and linker proteins, serving as the major light-harvesting complexes that capture and transfer light energy to photosystem II (PSII) and photosystem I (PSI) in cyanobacteria and eukaryotic red algae. In cyanobacteria, a rod-type PBS that does not have a core is specifically connected to PSI by a linker protein CpcL to form a PSI-CpcL-PBS supercomplex. However, the mechanism of CpcL-PBS association to PSI remains unclear. Here, we report the cryoelectron microscopic structures of PSI-CpcL-PBS at 2.98 Å and CpcL-PBS at 2.93 Å resolution from a cyanobacterium sp. PCC 7120, respectively. CpcL-PBS is located on the stromal side of a PSI tetramer and exhibits a structure of three-layered PBS consisting of four linkers (CpcL, CpcC1, CpcC2, PecC) and 18 pairs of phycocyanin αβ monomers. The C-terminal transmembrane helix of CpcL inserts to the membrane and interacts with PsaA, PsaB, and PsaM of PSI at an interface I between two PSI monomers, enabling the formation of the PSI-CpcL-PBS supercomplex. The exact structure of protein subunits and arrangement of bilin and chlorophyll pigments are revealed, which provide a structural basis for the assembly of PSI-CpcL-PBS and possible excitation energy transfer pathways from antennas to PSI within this supercomplex, shedding light on the organization and attachment of CpcL-PBS in cyanobacterial thylakoids. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_65729.map.gz emd_65729.map.gz | 258.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-65729-v30.xml emd-65729-v30.xml emd-65729.xml emd-65729.xml | 18.7 KB 18.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_65729_fsc.xml emd_65729_fsc.xml | 16.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_65729.png emd_65729.png | 105.9 KB | ||
| Filedesc metadata |  emd-65729.cif.gz emd-65729.cif.gz | 5.9 KB | ||
| Others |  emd_65729_half_map_1.map.gz emd_65729_half_map_1.map.gz emd_65729_half_map_2.map.gz emd_65729_half_map_2.map.gz | 475 MB 475 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-65729 http://ftp.pdbj.org/pub/emdb/structures/EMD-65729 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-65729 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-65729 | HTTPS FTP |
-Related structure data
| Related structure data |  9w7kMC  9w4jC  9wd5C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_65729.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_65729.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.04 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_65729_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_65729_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : CpcL-PBS2
| Entire | Name: CpcL-PBS2 |
|---|---|
| Components |
|
-Supramolecule #1: CpcL-PBS2
| Supramolecule | Name: CpcL-PBS2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) |
-Macromolecule #1: Photosystem I-associated linker protein CpcL
| Macromolecule | Name: Photosystem I-associated linker protein CpcL / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) |
| Molecular weight | Theoretical: 22.267941 KDa |
| Sequence | String: ALPLLEYKPT TQNQRVQSFG TADVNEDTPY IYRLENANSP SEIEELIWAA YRQVFNEQEI LKFNRQIGLE TQLKNRSITV KDFIRGLAK SERFYQLVVT PNNNYRLVEM SLKRLLGRSP YNEEEKIAWS IQIASKGWGG FVDALIDSTE YEQAFGDNTV P YQRKRLTT DRPFSFTPRY GADYRDRAGI VRP UniProtKB: Photosystem I-associated linker protein CpcL |
-Macromolecule #2: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod
| Macromolecule | Name: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) |
| Molecular weight | Theoretical: 32.122891 KDa |
| Sequence | String: AITTAASRLG TEPFSDAPKV ELRPKASREE VESVIRAVYR HVLGNDYILA SERLVSAESL LRDGNLTVRE FVRSVAKSEL YKKKFFYNS FQTRLIELNY KHLLGRAPYD ESEVVYHLDL YQNKGYDAEI DSYIDSWEYQ SNFGDNVVPY YRGFETQVGQ K TAGFNRIF ...String: AITTAASRLG TEPFSDAPKV ELRPKASREE VESVIRAVYR HVLGNDYILA SERLVSAESL LRDGNLTVRE FVRSVAKSEL YKKKFFYNS FQTRLIELNY KHLLGRAPYD ESEVVYHLDL YQNKGYDAEI DSYIDSWEYQ SNFGDNVVPY YRGFETQVGQ K TAGFNRIF RLYRGYANSD RAQVEGTKSR LARELASNKA STIVGPSGTN DSWGFRASAD VAPKKNLGNA VGEGDRVYRL EV TGIRSPG YPSVRRSSTV FIVPYERLSD KIQQVHKQGG KIVSVTSA UniProtKB: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod |
-Macromolecule #3: C-phycocyanin alpha subunit
| Macromolecule | Name: C-phycocyanin alpha subunit / type: protein_or_peptide / ID: 3 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) |
| Molecular weight | Theoretical: 17.344256 KDa |
| Sequence | String: VKTPITEAIA AADTQGRFLG NTELQSARGR YERAAASLEA ARGLTSNAQR LIDGATQAVY QKFPYTTQTP GPQFAADSRG KSKCARDVG HYLRIITYSL VAGGTGPLDE YLIAGLAEIN STFDLSPSWY VEALKHIKAN HGLSGQAANE ANTYIDYAIN A LS UniProtKB: C-phycocyanin alpha subunit |
-Macromolecule #4: C-phycocyanin beta subunit
| Macromolecule | Name: C-phycocyanin beta subunit / type: protein_or_peptide / ID: 4 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Nostoc sp. PCC 7120 = FACHB-418 (bacteria) Nostoc sp. PCC 7120 = FACHB-418 (bacteria) |
| Molecular weight | Theoretical: 18.272562 KDa |
| Sequence | String: TLDVFTKVVS QADSRGEFLS NEQLDALANV VKEGNKRLDV VNRITSNASA IVTNAARALF EEQPQLIAPG GNAYTNRRMA ACLRDMEII LRYVTYAILA GDASVLDDRC LNGLRETYQA LGTPGSSVAV GVQKMKDAAV GIANDPNGIT KGDCSQLISE V ASYFDRAA AAVG UniProtKB: C-phycocyanin beta subunit |
-Macromolecule #5: PHYCOCYANOBILIN
| Macromolecule | Name: PHYCOCYANOBILIN / type: ligand / ID: 5 / Number of copies: 36 / Formula: CYC |
|---|---|
| Molecular weight | Theoretical: 588.694 Da |
| Chemical component information | 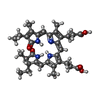 ChemComp-CYC: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOCONTINUUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





































