[English] 日本語
 Yorodumi
Yorodumi- EMDB-6067: Negative stain EM structure of the yeast dynein motor domain with... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6067 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Negative stain EM structure of the yeast dynein motor domain with stalk and MTBD in the presence of ATP | |||||||||
 Map data Map data | Reconstruction of yeast dynein motor domain with full stalk and MTBD in the presence of 5 mM ATP | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | motor proteins / power stroke / dynein / AAA protein / ATPase | |||||||||
| Function / homology |  Function and homology information Function and homology informationkaryogamy / nuclear migration along microtubule / astral microtubule / establishment of mitotic spindle localization / spindle pole body / minus-end-directed microtubule motor activity / dynein light intermediate chain binding / cytoplasmic dynein complex / nuclear migration / establishment of mitotic spindle orientation ...karyogamy / nuclear migration along microtubule / astral microtubule / establishment of mitotic spindle localization / spindle pole body / minus-end-directed microtubule motor activity / dynein light intermediate chain binding / cytoplasmic dynein complex / nuclear migration / establishment of mitotic spindle orientation / dynein intermediate chain binding / mitotic sister chromatid segregation / cytoplasmic microtubule / cytoplasmic microtubule organization / Neutrophil degranulation / mitotic spindle organization / cell cortex / ATP binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 23.0 Å | |||||||||
 Authors Authors | Bhabha G / Moeller A / Liao M / Speir JA / Vale RD / Cheng Y | |||||||||
 Citation Citation |  Journal: Cell / Year: 2014 Journal: Cell / Year: 2014Title: Allosteric communication in the dynein motor domain. Authors: Gira Bhabha / Hui-Chun Cheng / Nan Zhang / Arne Moeller / Maofu Liao / Jeffrey A Speir / Yifan Cheng / Ronald D Vale /  Abstract: Dyneins power microtubule motility using ring-shaped, AAA-containing motor domains. Here, we report X-ray and electron microscopy (EM) structures of yeast dynein bound to different ATP analogs, which ...Dyneins power microtubule motility using ring-shaped, AAA-containing motor domains. Here, we report X-ray and electron microscopy (EM) structures of yeast dynein bound to different ATP analogs, which collectively provide insight into the roles of dynein's two major ATPase sites, AAA1 and AAA3, in the conformational change mechanism. ATP binding to AAA1 triggers a cascade of conformational changes that propagate to all six AAA domains and cause a large movement of the "linker," dynein's mechanical element. In contrast to the role of AAA1 in driving motility, nucleotide transitions in AAA3 gate the transmission of conformational changes between AAA1 and the linker, suggesting that AAA3 acts as a regulatory switch. Further structural and mutational studies also uncover a role for the linker in regulating the catalytic cycle of AAA1. Together, these results reveal how dynein's two major ATP-binding sites initiate and modulate conformational changes in the motor domain during motility. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6067.map.gz emd_6067.map.gz | 744.2 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6067-v30.xml emd-6067-v30.xml emd-6067.xml emd-6067.xml | 9.4 KB 9.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_6067.png emd_6067.png | 124.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6067 http://ftp.pdbj.org/pub/emdb/structures/EMD-6067 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6067 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6067 | HTTPS FTP |
-Related structure data
| Related structure data |  6047C  6048C  6049C  6050C  6051C 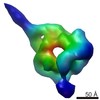 6052C 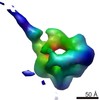 6053C 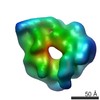 6054C  6055C 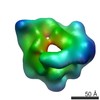 6056C  6058C  6059C  6060C  6061C  6062C  6063C  6064C  6065C  6066C  6068C  6069C  6070C  6071C  6072C  6073C  6074C  4w8fC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_6067.map.gz / Format: CCP4 / Size: 1001 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6067.map.gz / Format: CCP4 / Size: 1001 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Reconstruction of yeast dynein motor domain with full stalk and MTBD in the presence of 5 mM ATP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.26 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : yeast dynein motor domain with full stalk and MTBD in the presenc...
| Entire | Name: yeast dynein motor domain with full stalk and MTBD in the presence of 5 mM ATP |
|---|---|
| Components |
|
-Supramolecule #1000: yeast dynein motor domain with full stalk and MTBD in the presenc...
| Supramolecule | Name: yeast dynein motor domain with full stalk and MTBD in the presence of 5 mM ATP type: sample / ID: 1000 / Oligomeric state: monomer / Number unique components: 1 |
|---|---|
| Molecular weight | Theoretical: 303 KDa |
-Macromolecule #1: dynein
| Macromolecule | Name: dynein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 303 KDa |
| Recombinant expression | Organism:  |
| Sequence | UniProtKB: Dynein heavy chain, cytoplasmic |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.02 mg/mL |
|---|---|
| Buffer | pH: 8 Details: 50 mM Tris, pH 8.0, 200 mM sodium chloride, 1 mM EGTA, 2 mM magnesium acetate, 1 mM DTT |
| Staining | Type: NEGATIVE Details: Samples were applied to freshly glow discharged carbon coated grids and blotted off. Immediately after blotting, a 2% uranyl formate solution was applied for staining and blotted off. The ...Details: Samples were applied to freshly glow discharged carbon coated grids and blotted off. Immediately after blotting, a 2% uranyl formate solution was applied for staining and blotted off. The stain was applied five times per sample. Samples were allowed to air dry before imaging. |
| Grid | Details: Cu 400 mesh |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Date | Feb 27, 2013 |
| Image recording | Category: CCD / Film or detector model: TVIPS TEMCAM-F416 (4k x 4k) / Number real images: 168 / Average electron dose: 50 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 80000 |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 23.0 Å / Resolution method: OTHER / Software - Name: Relion / Number images used: 1827 |
|---|
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















