[English] 日本語
 Yorodumi
Yorodumi- EMDB-51939: Cryo-EM structure of the atovaquone-inhibited Complex III from th... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the atovaquone-inhibited Complex III from the Chlorocebus sabaeus respirasome | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | complex III / respirasome / mitochondria / green african monkey / atovaquone / ELECTRON TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationsubthalamus development / pons development / cerebellar Purkinje cell layer development / mitochondrial respiratory chain complex III assembly / pyramidal neuron development / thalamus development / respiratory chain complex III / quinol-cytochrome-c reductase / quinol-cytochrome-c reductase activity / mitochondrial electron transport, ubiquinol to cytochrome c ...subthalamus development / pons development / cerebellar Purkinje cell layer development / mitochondrial respiratory chain complex III assembly / pyramidal neuron development / thalamus development / respiratory chain complex III / quinol-cytochrome-c reductase / quinol-cytochrome-c reductase activity / mitochondrial electron transport, ubiquinol to cytochrome c / hypothalamus development / midbrain development / respiratory electron transport chain / hippocampus development / metalloendopeptidase activity / 2 iron, 2 sulfur cluster binding / electron transfer activity / mitochondrial inner membrane / heme binding / proteolysis / nucleoplasm / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Chlorocebus sabaeus (green monkey) Chlorocebus sabaeus (green monkey) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.67 Å | |||||||||
 Authors Authors | Maclean A / Muhleip A | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2025 Journal: Nat Struct Mol Biol / Year: 2025Title: Structure, assembly and inhibition of the Toxoplasma gondii respiratory chain supercomplex. Authors: Andrew E MacLean / Shikha Shikha / Mariana Ferreira Silva / Max J Gramelspacher / Aaron Nilsen / Katherine M Liebman / Sovitj Pou / Rolf W Winter / Amit Meir / Michael K Riscoe / J Stone ...Authors: Andrew E MacLean / Shikha Shikha / Mariana Ferreira Silva / Max J Gramelspacher / Aaron Nilsen / Katherine M Liebman / Sovitj Pou / Rolf W Winter / Amit Meir / Michael K Riscoe / J Stone Doggett / Lilach Sheiner / Alexander Mühleip /    Abstract: The apicomplexan mitochondrial electron transport chain is essential for parasite survival and displays a divergent subunit composition. Here we report cryo-electron microscopy structures of an ...The apicomplexan mitochondrial electron transport chain is essential for parasite survival and displays a divergent subunit composition. Here we report cryo-electron microscopy structures of an apicomplexan III-IV supercomplex and of the drug target complex III. The supercomplex structure reveals how clade-specific subunits form an apicomplexan-conserved III-IV interface with a unique, kinked architecture, suggesting that supercomplexes evolved independently in different eukaryotic lineages. A knockout resulting in supercomplex disassembly challenges the proposed role of III-IV in electron transfer efficiency as suggested for mammals. Nevertheless, knockout analysis indicates that III-IV is critical for parasite fitness. The complexes from the model parasite Toxoplasma gondii were inhibited with the antimalarial atovaquone, revealing interactions underpinning species specificity. They were also inhibited with endochin-like quinolone (ELQ)-300, an inhibitor in late-stage preclinical development. Notably, in the apicomplexan binding site, ELQ-300 is flipped compared with related compounds in the mammalian enzyme. On the basis of the binding modes and parasite-specific interactions discovered, we designed more potent ELQs with subnanomolar activity against T. gondii. Our findings reveal critical evolutionary differences in the role of supercomplexes in mitochondrial biology and provide insight into cytochrome b inhibition, informing future drug discovery. | |||||||||
| History |
|
- Structure visualization
Structure visualization
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_51939.map.gz emd_51939.map.gz | 777.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-51939-v30.xml emd-51939-v30.xml emd-51939.xml emd-51939.xml | 30.2 KB 30.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_51939_fsc.xml emd_51939_fsc.xml | 19.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_51939.png emd_51939.png | 145.9 KB | ||
| Masks |  emd_51939_msk_1.map emd_51939_msk_1.map | 824 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-51939.cif.gz emd-51939.cif.gz | 8.5 KB | ||
| Others |  emd_51939_half_map_1.map.gz emd_51939_half_map_1.map.gz emd_51939_half_map_2.map.gz emd_51939_half_map_2.map.gz | 764.4 MB 764.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-51939 http://ftp.pdbj.org/pub/emdb/structures/EMD-51939 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-51939 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-51939 | HTTPS FTP |
-Validation report
| Summary document |  emd_51939_validation.pdf.gz emd_51939_validation.pdf.gz | 990 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_51939_full_validation.pdf.gz emd_51939_full_validation.pdf.gz | 989.6 KB | Display | |
| Data in XML |  emd_51939_validation.xml.gz emd_51939_validation.xml.gz | 29.5 KB | Display | |
| Data in CIF |  emd_51939_validation.cif.gz emd_51939_validation.cif.gz | 39.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-51939 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-51939 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-51939 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-51939 | HTTPS FTP |
-Related structure data
| Related structure data |  9h8tMC  9g9tC  9i4xC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_51939.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_51939.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.98747 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
+Entire : mitochondrial respirasome from Chlorocebus sabaeus Vero cells inh...
+Supramolecule #1: mitochondrial respirasome from Chlorocebus sabaeus Vero cells inh...
+Macromolecule #1: Cytochrome c1
+Macromolecule #2: Cytochrome b-c1 complex subunit Rieske, mitochondrial
+Macromolecule #3: Cytochrome b-c1 complex subunit 7
+Macromolecule #4: Cytochrome b-c1 complex subunit 8
+Macromolecule #5: Complex III subunit 9
+Macromolecule #6: Cytochrome b-c1 complex subunit 6
+Macromolecule #7: Ubiquinol-cytochrome c reductase core protein 2
+Macromolecule #8: Cytochrome b
+Macromolecule #9: Ubiquinol-cytochrome c reductase core protein 1
+Macromolecule #10: UQCRB
+Macromolecule #11: HEME C
+Macromolecule #12: ZINC ION
+Macromolecule #13: FE2/S2 (INORGANIC) CLUSTER
+Macromolecule #14: CARDIOLIPIN
+Macromolecule #15: 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE
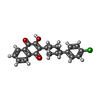
+Macromolecule #16: 2-[trans-4-(4-chlorophenyl)cyclohexyl]-3-hydroxynaphthalene-1,4-dione
+Macromolecule #17: 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine
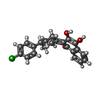
+Macromolecule #18: 2-[4-(4-CHLOROPHENYL)CYCLOHEXYLIDENE]-3,4-DIHYDROXY-1(2H)-NAPHTHA...
+Macromolecule #19: PROTOPORPHYRIN IX CONTAINING FE
+Macromolecule #20: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 301 K |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 36.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller