+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cdc45-GINS local refinement of human CMG G4 stall state | ||||||||||||
 Map data Map data | Cdc45-GINS local refinement of human CMG G4 stall state | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | helicase / DNA replication / cell division / fork stalling / translocation / REPLICATION / REPLICATION-DNA complex | ||||||||||||
| Biological species |  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.41 Å | ||||||||||||
 Authors Authors | Allwein B / Batra S / Remus D / Hite R | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Science / Year: 2025 Journal: Science / Year: 2025Title: G-quadruplex-stalled eukaryotic replisome structure reveals helical inchworm DNA translocation. Authors: Sahil Batra / Benjamin Allwein / Charanya Kumar / Sujan Devbhandari / Jan-Gert Brüning / Soon Bahng / Chong M Lee / Kenneth J Marians / Richard K Hite / Dirk Remus /  Abstract: DNA G-quadruplexes (G4s) are non-B-form DNA secondary structures that threaten genome stability by impeding DNA replication. To elucidate how G4s induce replication fork arrest, we characterized fork ...DNA G-quadruplexes (G4s) are non-B-form DNA secondary structures that threaten genome stability by impeding DNA replication. To elucidate how G4s induce replication fork arrest, we characterized fork collisions with preformed G4s in the parental DNA using reconstituted yeast and human replisomes. We demonstrate that a single G4 in the leading strand template is sufficient to stall replisomes by arresting the CMG helicase. Cryo-electron microscopy structures of stalled yeast and human CMG complexes reveal that the folded G4 is lodged inside the central CMG channel, arresting translocation. The G4 stabilizes the CMG at distinct translocation intermediates, suggesting an unprecedented helical inchworm mechanism for DNA translocation. These findings illuminate the eukaryotic replication fork mechanism under normal and perturbed conditions. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_47749.map.gz emd_47749.map.gz | 171.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-47749-v30.xml emd-47749-v30.xml emd-47749.xml emd-47749.xml | 22.9 KB 22.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_47749_fsc.xml emd_47749_fsc.xml | 14.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_47749.png emd_47749.png | 31.2 KB | ||
| Masks |  emd_47749_msk_1.map emd_47749_msk_1.map | 343 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-47749.cif.gz emd-47749.cif.gz | 5.1 KB | ||
| Others |  emd_47749_additional_1.map.gz emd_47749_additional_1.map.gz emd_47749_half_map_1.map.gz emd_47749_half_map_1.map.gz emd_47749_half_map_2.map.gz emd_47749_half_map_2.map.gz | 21 MB 318.2 MB 318.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-47749 http://ftp.pdbj.org/pub/emdb/structures/EMD-47749 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-47749 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-47749 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_47749.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_47749.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cdc45-GINS local refinement of human CMG G4 stall state | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.826 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_47749_msk_1.map emd_47749_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
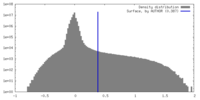
| Density Histograms |
-Additional map: Density-modified Cdc45-GINS local refinement of human CMG G4...
| File | emd_47749_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Density-modified Cdc45-GINS local refinement of human CMG G4 stall state | ||||||||||||
| Projections & Slices |
| ||||||||||||
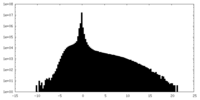
| Density Histograms |
-Half map: Cdc45-GINS local refinement half-map A of human CMG G4 stall state
| File | emd_47749_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cdc45-GINS local refinement half-map A of human CMG G4 stall state | ||||||||||||
| Projections & Slices |
| ||||||||||||
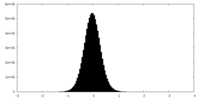
| Density Histograms |
-Half map: Cdc45-GINS local refinement half-map A of human CMG G4 stall state
| File | emd_47749_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cdc45-GINS local refinement half-map A of human CMG G4 stall state | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cdc45-GINS local refinement of human CMG G4 stall state
| Entire | Name: Cdc45-GINS local refinement of human CMG G4 stall state |
|---|---|
| Components |
|
-Supramolecule #1: Cdc45-GINS local refinement of human CMG G4 stall state
| Supramolecule | Name: Cdc45-GINS local refinement of human CMG G4 stall state type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#13 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 155.63 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | .01 mg/mL | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.6 Component:
| ||||||||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 400 / Support film - Material: GRAPHENE OXIDE / Support film - topology: CONTINUOUS / Support film - Film thickness: 0.5 | ||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: 30 second wait time after sample application; 30 second blot time, blot force 0. |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1 / Number real images: 3325 / Average exposure time: 3.0 sec. / Average electron dose: 66.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 29000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: Other / Chain - Initial model type: in silico model / Details: ModelAngelo |
|---|---|
| Details | Initial fitting was performed de novo by ModelAngelo, then iteratively improved with ChimeraX/ISOLDE, Coot, and Phenix real-space refinement algorithms. |
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Overall B value: 49.99 |
 Movie
Movie Controller
Controller





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)