[English] 日本語
 Yorodumi
Yorodumi- EMDB-4287: Human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound to... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4287 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound to 1 RBD domain from RPAP3 | ||||||||||||
 Map data Map data | Structure of a human R2TP subcomplex, comprising a RUVBL1-RUVBL2 hexamer bound to one RBD domain of RPAP3 | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | R2TP / HSP90 co-chaperone / PIH1D1 / RPAP3 / RUVBL1-RUVBL2 / CHAPERONE | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationpromoter-enhancer loop anchoring activity / telomerase RNA localization to Cajal body / establishment of protein localization to chromatin / regulation of DNA strand elongation / positive regulation of telomere maintenance in response to DNA damage / R2TP complex / dynein axonemal particle / RPAP3/R2TP/prefoldin-like complex / Swr1 complex / Ino80 complex ...promoter-enhancer loop anchoring activity / telomerase RNA localization to Cajal body / establishment of protein localization to chromatin / regulation of DNA strand elongation / positive regulation of telomere maintenance in response to DNA damage / R2TP complex / dynein axonemal particle / RPAP3/R2TP/prefoldin-like complex / Swr1 complex / Ino80 complex / regulation of double-strand break repair / box C/D snoRNP assembly / regulation of chromosome organization / NuA4 histone acetyltransferase complex / regulation of DNA replication / MLL1 complex / TFIID-class transcription factor complex binding / regulation of embryonic development / Telomere Extension By Telomerase / protein folding chaperone complex / regulation of DNA repair / RNA polymerase II core promoter sequence-specific DNA binding / positive regulation of double-strand break repair via homologous recombination / Deposition of new CENPA-containing nucleosomes at the centromere / telomere maintenance / TBP-class protein binding / ciliary tip / acrosomal vesicle / positive regulation of DNA repair / DNA helicase activity / cellular response to estradiol stimulus / euchromatin / negative regulation of canonical Wnt signaling pathway / Formation of the beta-catenin:TCF transactivating complex / ADP binding / chromatin DNA binding / beta-catenin binding / DNA Damage Recognition in GG-NER / nuclear matrix / cellular response to UV / : / transcription corepressor activity / UCH proteinases / positive regulation of canonical Wnt signaling pathway / nucleosome / HATs acetylate histones / ATPase binding / protein folding / DNA recombination / spermatogenesis / regulation of apoptotic process / DNA helicase / transcription coactivator activity / regulation of cell cycle / Ub-specific processing proteases / nuclear speck / protein stabilization / cilium / ciliary basal body / RNA polymerase II cis-regulatory region sequence-specific DNA binding / cadherin binding / chromatin remodeling / ribonucleoprotein complex / cell division / DNA repair / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / centrosome / positive regulation of DNA-templated transcription / protein homodimerization activity / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / extracellular exosome / nucleoplasm / ATP binding / membrane / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
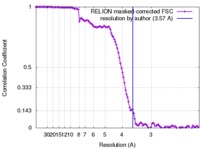
| Method | single particle reconstruction / cryo EM / Resolution: 3.57 Å | ||||||||||||
 Authors Authors | Martino F / Munoz-Hernandez H / Rodriguez CF / Pearl LH / Llorca O | ||||||||||||
| Funding support |  Spain, 3 items Spain, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2018 Journal: Nat Commun / Year: 2018Title: RPAP3 provides a flexible scaffold for coupling HSP90 to the human R2TP co-chaperone complex. Authors: Fabrizio Martino / Mohinder Pal / Hugo Muñoz-Hernández / Carlos F Rodríguez / Rafael Núñez-Ramírez / David Gil-Carton / Gianluca Degliesposti / J Mark Skehel / S Mark Roe / ...Authors: Fabrizio Martino / Mohinder Pal / Hugo Muñoz-Hernández / Carlos F Rodríguez / Rafael Núñez-Ramírez / David Gil-Carton / Gianluca Degliesposti / J Mark Skehel / S Mark Roe / Chrisostomos Prodromou / Laurence H Pearl / Oscar Llorca /   Abstract: The R2TP/Prefoldin-like co-chaperone, in concert with HSP90, facilitates assembly and cellular stability of RNA polymerase II, and complexes of PI3-kinase-like kinases such as mTOR. However, the ...The R2TP/Prefoldin-like co-chaperone, in concert with HSP90, facilitates assembly and cellular stability of RNA polymerase II, and complexes of PI3-kinase-like kinases such as mTOR. However, the mechanism by which this occurs is poorly understood. Here we use cryo-EM and biochemical studies on the human R2TP core (RUVBL1-RUVBL2-RPAP3-PIH1D1) which reveal the distinctive role of RPAP3, distinguishing metazoan R2TP from the smaller yeast equivalent. RPAP3 spans both faces of a single RUVBL ring, providing an extended scaffold that recruits clients and provides a flexible tether for HSP90. A 3.6 Å cryo-EM structure reveals direct interaction of a C-terminal domain of RPAP3 and the ATPase domain of RUVBL2, necessary for human R2TP assembly but absent from yeast. The mobile TPR domains of RPAP3 map to the opposite face of the ring, associating with PIH1D1, which mediates client protein recruitment. Thus, RPAP3 provides a flexible platform for bringing HSP90 into proximity with diverse client proteins. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4287.map.gz emd_4287.map.gz | 7.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4287-v30.xml emd-4287-v30.xml emd-4287.xml emd-4287.xml | 23.2 KB 23.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4287_fsc.xml emd_4287_fsc.xml | 11.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_4287.png emd_4287.png | 58.6 KB | ||
| Filedesc metadata |  emd-4287.cif.gz emd-4287.cif.gz | 7.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4287 http://ftp.pdbj.org/pub/emdb/structures/EMD-4287 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4287 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4287 | HTTPS FTP |
-Related structure data
| Related structure data |  6fo1MC  4289C  4290C  4291C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4287.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4287.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of a human R2TP subcomplex, comprising a RUVBL1-RUVBL2 hexamer bound to one RBD domain of RPAP3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : a human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound ...
| Entire | Name: a human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound to 1 RBD domain of RPAP3 |
|---|---|
| Components |
|
-Supramolecule #1: a human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound ...
| Supramolecule | Name: a human R2TP subcomplex containing 1 RUVBL1-RUVBL2 hexamer bound to 1 RBD domain of RPAP3 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: RuvB-like 1
| Macromolecule | Name: RuvB-like 1 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 50.296914 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKIEEVKSTT KTQRIASHSH VKGLGLDESG LAKQAASGLV GQENAREACG VIVELIKSKK MAGRAVLLAG PPGTGKTALA LAIAQELGS KVPFCPMVGS EVYSTEIKKT EVLMENFRRA IGLRIKETKE VYEGEVTELT PCETENPMGG YGKTISHVII G LKTAKGTK ...String: MKIEEVKSTT KTQRIASHSH VKGLGLDESG LAKQAASGLV GQENAREACG VIVELIKSKK MAGRAVLLAG PPGTGKTALA LAIAQELGS KVPFCPMVGS EVYSTEIKKT EVLMENFRRA IGLRIKETKE VYEGEVTELT PCETENPMGG YGKTISHVII G LKTAKGTK QLKLDPSIFE SLQKERVEAG DVIYIEANSG AVKRQGRCDT YATEFDLEAE EYVPLPKGDV HKKKEIIQDV TL HDLDVAN ARPQGGQDIL SMMGQLMKPK KTEITDKLRG EINKVVNKYI DQGIAELVPG VLFVDEVHML DIECFTYLHR ALE SSIAPI VIFASNRGNC VIRGTEDITS PHGIPLDLLD RVMIIRTMLY TPQEMKQIIK IRAQTEGINI SEEALNHLGE IGTK TTLRY SVQLLTPANL LAKINGKDSI EKEHVEEISE LFYDAKSSAK ILADQQDKYM K UniProtKB: RuvB-like 1 |
-Macromolecule #2: RuvB-like 2
| Macromolecule | Name: RuvB-like 2 / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 51.222465 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MATVTATTKV PEIRDVTRIE RIGAHSHIRG LGLDDALEPR QASQGMVGQL AARRAAGVVL EMIREGKIAG RAVLIAGQPG TGKTAIAMG MAQALGPDTP FTAIAGSEIF SLEMSKTEAL TQAFRRSIGV RIKEETEIIE GEVVEIQIDR PATGTGSKVG K LTLKTTEM ...String: MATVTATTKV PEIRDVTRIE RIGAHSHIRG LGLDDALEPR QASQGMVGQL AARRAAGVVL EMIREGKIAG RAVLIAGQPG TGKTAIAMG MAQALGPDTP FTAIAGSEIF SLEMSKTEAL TQAFRRSIGV RIKEETEIIE GEVVEIQIDR PATGTGSKVG K LTLKTTEM ETIYDLGTKM IESLTKDKVQ AGDVITIDKA TGKISKLGRS FTRARDYDAM GSQTKFVQCP DGELQKRKEV VH TVSLHEI DVINSRTQGF LALFSGDTGE IKSEVREQIN AKVAEWREEG KAEIIPGVLF IDEVHMLDIE SFSFLNRALE SDM APVLIM ATNRGITRIR GTSYQSPHGI PIDLLDRLLI VSTTPYSEKD TKQILRIRCE EEDVEMSEDA YTVLTRIGLE TSLR YAIQL ITAASLVCRK RKGTEVQVDD IKRVYSLFLD ESRSTQYMKE YQDAFLFNEL KGETMDTS UniProtKB: RuvB-like 2 |
-Macromolecule #3: RNA polymerase II-associated protein 3
| Macromolecule | Name: RNA polymerase II-associated protein 3 / type: protein_or_peptide / ID: 3 Details: MTSANKAIELQLQVKQNAEELQDFMRDLENWEKDIKQKDMELRRQNGVPEENLPPIRNGN FRKKKKGKAKESSKKTREENTKNRIKSYDYEAWAKLDVDRILDELDKDDSTHESLSQESE SEEDGIHVDSQKALVLKEKGNKYFKQGKYDEAIDCYTKGMDADPYNPVLPTNRASAYFRL ...Details: MTSANKAIELQLQVKQNAEELQDFMRDLENWEKDIKQKDMELRRQNGVPEENLPPIRNGN FRKKKKGKAKESSKKTREENTKNRIKSYDYEAWAKLDVDRILDELDKDDSTHESLSQESE SEEDGIHVDSQKALVLKEKGNKYFKQGKYDEAIDCYTKGMDADPYNPVLPTNRASAYFRL KKFAVAESDCNLAVALNRSYTKAYSRRGAARFALQKLEEAKKDYERVLELEPNNFEATNE LRKISQALASKENSYPKEADIVIKSTEGERKQIEAQQNKQQAISEKDRGNGFFKEGKYER AIECYTRGIAADGANALLPANRAMAYLKIQKYEEAEKDCTQAILLDGSYSKAFARRGTAR TFLGKLNEAKQDFETVLLLEPGNKQAVTELSKIKKELIEKGHWDDVFLDSTQRQNVVKPI DNPPHPGSTKPLKKVIIEETGNLIQTIDVPDSTTAAAPENNPINLANVIAATGTTSKKNS SQDDLFPTSDTPRAKVLKIEEVSDTSSLQPQASLKQDVCQSYSEKMPIEIEQKPAQFATT VLPPIPANSFQLESDFRQLKSSPDMLYQYLKQIEPSLYPKLFQKNLDPDVFNQIVKILHD FYIEKEKPLLIFEILQRLSELKRFDMAVMFMSETEKKIARALFNHIDKSGLKDSSVEELK KRYGG Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 75.830547 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTSANKAIEL QLQVKQNAEE LQDFMRDLEN WEKDIKQKDM ELRRQNGVPE ENLPPIRNGN FRKKKKGKAK ESSKKTREEN TKNRIKSYD YEAWAKLDVD RILDELDKDD STHESLSQES ESEEDGIHVD SQKALVLKEK GNKYFKQGKY DEAIDCYTKG M DADPYNPV ...String: MTSANKAIEL QLQVKQNAEE LQDFMRDLEN WEKDIKQKDM ELRRQNGVPE ENLPPIRNGN FRKKKKGKAK ESSKKTREEN TKNRIKSYD YEAWAKLDVD RILDELDKDD STHESLSQES ESEEDGIHVD SQKALVLKEK GNKYFKQGKY DEAIDCYTKG M DADPYNPV LPTNRASAYF RLKKFAVAES DCNLAVALNR SYTKAYSRRG AARFALQKLE EAKKDYERVL ELEPNNFEAT NE LRKISQA LASKENSYPK EADIVIKSTE GERKQIEAQQ NKQQAISEKD RGNGFFKEGK YERAIECYTR GIAADGANAL LPA NRAMAY LKIQKYEEAE KDCTQAILLD GSYSKAFARR GTARTFLGKL NEAKQDFETV LLLEPGNKQA VTELSKIKKE LIEK GHWDD VFLDSTQRQN VVKPIDNPPH PGSTKPLKKV IIEETGNLIQ TIDVPDSTTA AAPENNPINL ANVIAATGTT SKKNS SQDD LFPTSDTPRA KVLKIEEVSD TSSLQPQASL KQDVCQSYSE KMPIEIEQKP AQFATTVLPP IPANSFQLES DFRQLK SSP DMLYQYLKQI EPSLYPKLFQ KNLDPDVFNQ IVKILHDFYI EKEKPLLIFE ILQRLSELKR FDMAVMFMSE TEKKIAR AL FNHIDKSGLK DSSVEELKKR YGG UniProtKB: RNA polymerase II-associated protein 3 |
-Macromolecule #4: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 4 / Number of copies: 6 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.8 Component:
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 52.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-6fo1: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)