+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | momSalB bound Kappa Opioid Receptor in complex with GoA | |||||||||
 Map data Map data | The locally-refined map of kappa-opioid receptor region | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / Receptor / Kappa / KOR / Opioid / MEMBRANE PROTEIN | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||
 Authors Authors | Fay JF / Che T | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Ligand and G-protein selectivity in the κ-opioid receptor. Authors: Jianming Han / Jingying Zhang / Antonina L Nazarova / Sarah M Bernhard / Brian E Krumm / Lei Zhao / Jordy Homing Lam / Vipin A Rangari / Susruta Majumdar / David E Nichols / Vsevolod ...Authors: Jianming Han / Jingying Zhang / Antonina L Nazarova / Sarah M Bernhard / Brian E Krumm / Lei Zhao / Jordy Homing Lam / Vipin A Rangari / Susruta Majumdar / David E Nichols / Vsevolod Katritch / Peng Yuan / Jonathan F Fay / Tao Che /  Abstract: The κ-opioid receptor (KOR) represents a highly desirable therapeutic target for treating not only pain but also addiction and affective disorders. However, the development of KOR analgesics has ...The κ-opioid receptor (KOR) represents a highly desirable therapeutic target for treating not only pain but also addiction and affective disorders. However, the development of KOR analgesics has been hindered by the associated hallucinogenic side effects. The initiation of KOR signalling requires the G-family proteins including the conventional (G, G, G, G and G) and nonconventional (G and G) subtypes. How hallucinogens exert their actions through KOR and how KOR determines G-protein subtype selectivity are not well understood. Here we determined the active-state structures of KOR in a complex with multiple G-protein heterotrimers-G, G, G and G-using cryo-electron microscopy. The KOR-G-protein complexes are bound to hallucinogenic salvinorins or highly selective KOR agonists. Comparisons of these structures reveal molecular determinants critical for KOR-G-protein interactions as well as key elements governing G-family subtype selectivity and KOR ligand selectivity. Furthermore, the four G-protein subtypes display an intrinsically different binding affinity and allosteric activity on agonist binding at KOR. These results provide insights into the actions of opioids and G-protein-coupling specificity at KOR and establish a foundation to examine the therapeutic potential of pathway-selective agonists of KOR. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41876.map.gz emd_41876.map.gz | 84.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41876-v30.xml emd-41876-v30.xml emd-41876.xml emd-41876.xml | 14 KB 14 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41876.png emd_41876.png | 46.9 KB | ||
| Filedesc metadata |  emd-41876.cif.gz emd-41876.cif.gz | 4.8 KB | ||
| Others |  emd_41876_half_map_1.map.gz emd_41876_half_map_1.map.gz emd_41876_half_map_2.map.gz emd_41876_half_map_2.map.gz | 84.4 MB 84.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41876 http://ftp.pdbj.org/pub/emdb/structures/EMD-41876 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41876 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41876 | HTTPS FTP |
-Validation report
| Summary document |  emd_41876_validation.pdf.gz emd_41876_validation.pdf.gz | 774.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41876_full_validation.pdf.gz emd_41876_full_validation.pdf.gz | 774.2 KB | Display | |
| Data in XML |  emd_41876_validation.xml.gz emd_41876_validation.xml.gz | 12.8 KB | Display | |
| Data in CIF |  emd_41876_validation.cif.gz emd_41876_validation.cif.gz | 15 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41876 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41876 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41876 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41876 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41876.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41876.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The locally-refined map of kappa-opioid receptor region | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.88 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half map of the locally-refined map of kappa-opioid receptor region
| File | emd_41876_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of the locally-refined map of kappa-opioid receptor region | ||||||||||||
| Projections & Slices |
| ||||||||||||
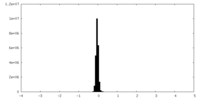
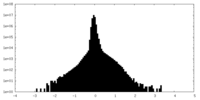
| Density Histograms |
-Half map: Half map of the locally-refined map of kappa-opioid receptor region
| File | emd_41876_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of the locally-refined map of kappa-opioid receptor region | ||||||||||||
| Projections & Slices |
| ||||||||||||
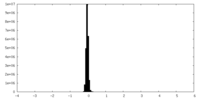
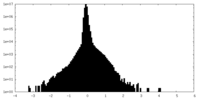
| Density Histograms |
- Sample components
Sample components
-Entire : momSalB bound Kappa Opioid Receptor in complex with GoA
| Entire | Name: momSalB bound Kappa Opioid Receptor in complex with GoA |
|---|---|
| Components |
|
-Supramolecule #1: momSalB bound Kappa Opioid Receptor in complex with GoA
| Supramolecule | Name: momSalB bound Kappa Opioid Receptor in complex with GoA type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Kappa-type opioid receptor
| Macromolecule | Name: Kappa-type opioid receptor / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
| Sequence | String: LGSISPAIPV IITAVYSVVF VVGLVGNSLV MFVIIRYTKM KTATNIYIFN LALADALVTT TMPFQSTVYL MNSWPFGDVL CKIVLSIDY YNMFTSIFTL TMMSVDRYIA VCHPVKALDF RTPLKAKIIN ICIWLLSSSV GISAIVLGGT KVREDVDVIE C SLQFPDDD ...String: LGSISPAIPV IITAVYSVVF VVGLVGNSLV MFVIIRYTKM KTATNIYIFN LALADALVTT TMPFQSTVYL MNSWPFGDVL CKIVLSIDY YNMFTSIFTL TMMSVDRYIA VCHPVKALDF RTPLKAKIIN ICIWLLSSSV GISAIVLGGT KVREDVDVIE C SLQFPDDD YSWWDLFMKI CVFIFAFVIP VLIIIVCYTL MILRLKSVRL LSGSREKDRN LRRITRLVLV VVAVFVVCWT PI HIFILVE ALGSTSHSTA ALSSYYFCIA LGYTNSSLNP ILYAFLDENF KRCFRDFCFP LKMRMERQST S |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 3853 / Average electron dose: 42.7 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.7 µm / Nominal defocus min: 0.2 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 991076 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)