[English] 日本語
 Yorodumi
Yorodumi- EMDB-3970: The in situ structure of the Chlamydomonas COPI coat: average fro... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3970 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
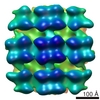
| Title | The in situ structure of the Chlamydomonas COPI coat: average from vesicles | |||||||||
 Map data Map data | The in situ structure of the Chlamydomonas COPI coat derived from budded vesicles. | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | subtomogram averaging / cryo EM / Resolution: 23.0 Å | |||||||||
 Authors Authors | Bykov YS / Schaffer M / Dodonova SO / Albert S / Plitzko JM / Baumeister W / Engel BD / Briggs JAG | |||||||||
 Citation Citation |  Journal: Elife / Year: 2017 Journal: Elife / Year: 2017Title: The structure of the COPI coat determined within the cell. Authors: Yury S Bykov / Miroslava Schaffer / Svetlana O Dodonova / Sahradha Albert / Jürgen M Plitzko / Wolfgang Baumeister / Benjamin D Engel / John Ag Briggs /   Abstract: COPI-coated vesicles mediate trafficking within the Golgi apparatus and from the Golgi to the endoplasmic reticulum. The structures of membrane protein coats, including COPI, have been extensively ...COPI-coated vesicles mediate trafficking within the Golgi apparatus and from the Golgi to the endoplasmic reticulum. The structures of membrane protein coats, including COPI, have been extensively studied with reconstitution systems using purified components. Previously we have determined a complete structural model of the reconstituted COPI coat (Dodonova et al., 2017). Here, we applied cryo-focused ion beam milling, cryo-electron tomography and subtomogram averaging to determine the native structure of the COPI coat within vitrified cells. The native algal structure resembles the mammalian structure, but additionally reveals cargo bound beneath β'-COP. We find that all coat components disassemble simultaneously and relatively rapidly after budding. Structural analysis , maintaining Golgi topology, shows that vesicles change their size, membrane thickness, and cargo content as they progress from to , but the structure of the coat machinery remains constant. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3970.map.gz emd_3970.map.gz | 7.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3970-v30.xml emd-3970-v30.xml emd-3970.xml emd-3970.xml | 11.5 KB 11.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_3970.png emd_3970.png | 94.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3970 http://ftp.pdbj.org/pub/emdb/structures/EMD-3970 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3970 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3970 | HTTPS FTP |
-Related structure data
| Related structure data |  3968C  3969C  3971C  3972C  3973C  3974C  3975C  3976C  3977C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_3970.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3970.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The in situ structure of the Chlamydomonas COPI coat derived from budded vesicles. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.42 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Whole Chlamydomonas cells
| Entire | Name: Whole Chlamydomonas cells |
|---|---|
| Components |
|
-Supramolecule #1: Whole Chlamydomonas cells
| Supramolecule | Name: Whole Chlamydomonas cells / type: organelle_or_cellular_component / ID: 1 / Parent: 0 Details: Grown suspended in TAP media, with normal atmosphere aeration and constant light |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | cell |
- Sample preparation
Sample preparation
| Buffer | pH: 7 / Details: TAP media |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 90 % / Instrument: FEI VITROBOT MARK IV Details: Blotted from the back side for 10 seconds with 10 blot force before plunging. |
| Details | The cells were frozen onto grids, then thinned using cryo-focused ion beam milling. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Lower energy threshold: 0 eV / Energy filter - Upper energy threshold: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Frames/image: 1-12 / Average exposure time: 1.5 sec. / Average electron dose: 1.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 6.0 µm / Nominal defocus min: 4.0 µm / Nominal magnification: 42000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Point group: C3 (3 fold cyclic) / Algorithm: BACK PROJECTION / Resolution.type: BY AUTHOR / Resolution: 23.0 Å / Resolution method: FSC 0.143 CUT-OFF / Software: (Name: TOM, AV3) Details: For this average only subtomograms found in budded vesicles were used. No additional alignment was performed. Number subtomograms used: 1615 |
|---|---|
| Extraction | Number tomograms: 29 / Number images used: 53109 / Reference model: None / Software: (Name: TOM, AV3) Details: Subtomograms were extracted uniformly around the membrane surface with initial angles normal to it. |
| CTF correction | Software: (Name: CTFFIND, CTFPHASEFLIP) |
| Final angle assignment | Type: NOT APPLICABLE / Software - Name: AV3 |
-Atomic model buiding 1
| Refinement | Protocol: RIGID BODY FIT |
|---|
 Movie
Movie Controller
Controller



 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















