[English] 日本語
 Yorodumi
Yorodumi- EMDB-38788: Mycobacterium smegmatis 50S ribosomal subunit with Erythromycin -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
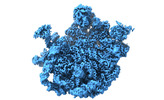
| Title | Mycobacterium smegmatis 50S ribosomal subunit with Erythromycin | |||||||||
 Map data Map data | Mycobacterium smegmatis 50S ribosomal subunit in complex with erythromycin | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Antibiotic / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationlarge ribosomal subunit / transferase activity / 5S rRNA binding / ribosomal large subunit assembly / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / negative regulation of translation / rRNA binding ...large ribosomal subunit / transferase activity / 5S rRNA binding / ribosomal large subunit assembly / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / negative regulation of translation / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / mRNA binding / metal ion binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) | |||||||||
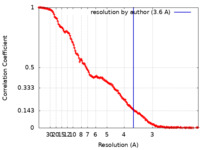
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Srinivasan K / Banerjee A / Sengupta J | |||||||||
| Funding support |  India, 2 items India, 2 items
| |||||||||
 Citation Citation |  Journal: Structure / Year: 2024 Journal: Structure / Year: 2024Title: Cryo-EM structures reveal the molecular mechanism of HflX-mediated erythromycin resistance in mycobacteria. Authors: Krishnamoorthi Srinivasan / Aneek Banerjee / Jayati Sengupta /  Abstract: Mycobacterial HflX confers resistance against macrolide antibiotics. However, the exact molecular mechanism is poorly understood. To gain further insights, we determined the cryo-EM structures of M. ...Mycobacterial HflX confers resistance against macrolide antibiotics. However, the exact molecular mechanism is poorly understood. To gain further insights, we determined the cryo-EM structures of M. smegmatis (Msm) HflX-50S subunit and 50S subunit-erythromycin (ERY) complexes at a global resolution of approximately 3 Å. A conserved nucleotide A2286 at the gate of nascent peptide exit tunnel (NPET) adopts a swayed conformation in HflX-50S complex and interacts with a loop within the linker helical (LH) domain of MsmHflX that contains an additional 9 residues insertion. Interestingly, the swaying of this nucleotide, which is usually found in the non-swayed conformation, is induced by erythromycin binding. Furthermore, we observed that erythromycin decreases HflX's ribosome-dependent GTP hydrolysis, resulting in its enhanced binding and anti-association activity on the 50S subunit. Our findings reveal how mycobacterial HflX senses the presence of macrolides at the peptide tunnel entrance and confers antibiotic resistance in mycobacteria. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38788.map.gz emd_38788.map.gz | 255.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38788-v30.xml emd-38788-v30.xml emd-38788.xml emd-38788.xml | 54.5 KB 54.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_38788_fsc.xml emd_38788_fsc.xml | 19.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_38788.png emd_38788.png | 96.2 KB | ||
| Filedesc metadata |  emd-38788.cif.gz emd-38788.cif.gz | 10.3 KB | ||
| Others |  emd_38788_half_map_1.map.gz emd_38788_half_map_1.map.gz emd_38788_half_map_2.map.gz emd_38788_half_map_2.map.gz | 225.2 MB 225.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38788 http://ftp.pdbj.org/pub/emdb/structures/EMD-38788 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38788 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38788 | HTTPS FTP |
-Related structure data
| Related structure data |  8xz3MC  8kabC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_38788.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38788.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Mycobacterium smegmatis 50S ribosomal subunit in complex with erythromycin | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
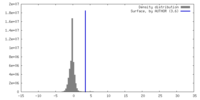
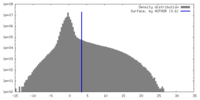
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half-map of Mycobacterium smegmatis 50S ribosomal subunit in...
| File | emd_38788_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map of Mycobacterium smegmatis 50S ribosomal subunit in complex with erythromycin | ||||||||||||
| Projections & Slices |
| ||||||||||||
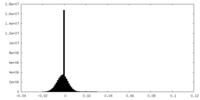
| Density Histograms |
-Half map: Half-map of Mycobacterium smegmatis 50S ribosomal subunit in...
| File | emd_38788_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map of Mycobacterium smegmatis 50S ribosomal subunit in complex with erythromycin | ||||||||||||
| Projections & Slices |
| ||||||||||||
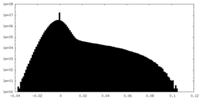
| Density Histograms |
- Sample components
Sample components
+Entire : Mycobacterium smegmatis 50S ribosomal subunit-erythromycin complex
+Supramolecule #1: Mycobacterium smegmatis 50S ribosomal subunit-erythromycin complex
+Supramolecule #2: Mycobacterium smegmatis 50S ribosomal subunit
+Macromolecule #1: 50S ribosomal protein bL37
+Macromolecule #4: Large ribosomal subunit protein uL2
+Macromolecule #5: Large ribosomal subunit protein uL3
+Macromolecule #6: Large ribosomal subunit protein uL4
+Macromolecule #7: Large ribosomal subunit protein uL5
+Macromolecule #8: Large ribosomal subunit protein uL6
+Macromolecule #9: 50S ribosomal protein L9
+Macromolecule #10: Large ribosomal subunit protein uL10
+Macromolecule #11: Large ribosomal subunit protein uL11
+Macromolecule #12: Large ribosomal subunit protein uL13
+Macromolecule #13: 50S ribosomal protein L14
+Macromolecule #14: Large ribosomal subunit protein uL15
+Macromolecule #15: Large ribosomal subunit protein uL16
+Macromolecule #16: Large ribosomal subunit protein bL17
+Macromolecule #17: Large ribosomal subunit protein uL18
+Macromolecule #18: 50S ribosomal protein L19
+Macromolecule #19: Large ribosomal subunit protein bL20
+Macromolecule #20: Large ribosomal subunit protein bL21
+Macromolecule #21: Large ribosomal subunit protein uL22
+Macromolecule #22: Large ribosomal subunit protein uL23
+Macromolecule #23: Large ribosomal subunit protein uL24
+Macromolecule #24: Large ribosomal subunit protein bL25
+Macromolecule #25: Large ribosomal subunit protein bL27
+Macromolecule #26: Large ribosomal subunit protein bL28
+Macromolecule #27: Large ribosomal subunit protein uL29
+Macromolecule #28: Large ribosomal subunit protein uL30
+Macromolecule #29: Large ribosomal subunit protein bL32
+Macromolecule #30: Large ribosomal subunit protein bL33A
+Macromolecule #31: Large ribosomal subunit protein bL34
+Macromolecule #32: Large ribosomal subunit protein bL35
+Macromolecule #33: 50S ribosomal protein L36
+Macromolecule #34: Large ribosomal subunit protein bL31
+Macromolecule #2: 23S rRNA
+Macromolecule #3: 5S rRNA
+Macromolecule #35: ERYTHROMYCIN A
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 28.75 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.3000000000000003 µm / Nominal defocus min: 1.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)