+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MRE-269 bound Prostacyclin Receptor G protein complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Agonist / Complex / lipid receptor / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationprostacyclin receptor activity / Prostanoid ligand receptors / negative regulation of platelet-derived growth factor receptor signaling pathway / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / guanyl-nucleotide exchange factor activity / negative regulation of smooth muscle cell proliferation / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta ...prostacyclin receptor activity / Prostanoid ligand receptors / negative regulation of platelet-derived growth factor receptor signaling pathway / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / guanyl-nucleotide exchange factor activity / negative regulation of smooth muscle cell proliferation / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G protein-coupled acetylcholine receptor signaling pathway / G-protein activation / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through CDC42 / Glucagon signaling in metabolic regulation / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / photoreceptor disc membrane / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / ADP signalling through P2Y purinoceptor 1 / cellular response to catecholamine stimulus / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / adenylate cyclase-activating dopamine receptor signaling pathway / GPER1 signaling / Inactivation, recovery and regulation of the phototransduction cascade / cellular response to prostaglandin E stimulus / G-protein beta-subunit binding / heterotrimeric G-protein complex / cell-cell signaling / G alpha (12/13) signalling events / sensory perception of taste / extracellular vesicle / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / retina development in camera-type eye / positive regulation of cytosolic calcium ion concentration / GTPase binding / Ca2+ pathway / fibroblast proliferation / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / G alpha (i) signalling events / response to lipopolysaccharide / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (q) signalling events / Ras protein signal transduction / Extra-nuclear estrogen signaling / cell population proliferation / G protein-coupled receptor signaling pathway / inflammatory response / lysosomal membrane / GTPase activity / synapse / GTP binding / protein-containing complex binding / signal transduction / extracellular exosome / metal ion binding / membrane / plasma membrane / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.41 Å | |||||||||
 Authors Authors | Wang JJ / Jin S / Zhang H / Xu Y / Hu W / Jiang Y / Chen C / Wang DW / Xu HE / Wu C | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2024 Journal: Sci Adv / Year: 2024Title: Molecular recognition and activation of the prostacyclin receptor by anti-pulmonary arterial hypertension drugs. Authors: James Jiqi Wang / Sanshan Jin / Heng Zhang / Youwei Xu / Wen Hu / Yi Jiang / Chen Chen / Dao Wen Wang / H Eric Xu / Canrong Wu /  Abstract: The prostacyclin (PGI) receptor (IP) is a G-coupled receptor associated with blood pressure regulation, allergy, and inflammatory response. It is a main therapeutic target for pulmonary arterial ...The prostacyclin (PGI) receptor (IP) is a G-coupled receptor associated with blood pressure regulation, allergy, and inflammatory response. It is a main therapeutic target for pulmonary arterial hypertension (PAH) and several other diseases. Here we report cryo-electron microscopy (cryo-EM) structures of the human IP-G complex bound with two anti-PAH drugs, treprostinil and MRE-269 (active form of selexipag), at global resolutions of 2.56 and 2.41 angstrom, respectively. These structures revealed distinct features governing IP ligand binding, receptor activation, and G protein coupling. Moreover, comparison of the activated IP structures uncovered the mechanism and key residues that determine the superior selectivity of MRE-269 over treprostinil. Combined with molecular docking and functional studies, our structures provide insight into agonist selectivity, ligand recognition, receptor activation, and G protein coupling. Our results provide a structural template for further improving IP-targeting drugs to reduce off-target activation of prostanoid receptors and adverse effects. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38095.map.gz emd_38095.map.gz | 115.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38095-v30.xml emd-38095-v30.xml emd-38095.xml emd-38095.xml | 21.3 KB 21.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_38095.png emd_38095.png | 51.3 KB | ||
| Filedesc metadata |  emd-38095.cif.gz emd-38095.cif.gz | 6.5 KB | ||
| Others |  emd_38095_additional_1.map.gz emd_38095_additional_1.map.gz emd_38095_half_map_1.map.gz emd_38095_half_map_1.map.gz emd_38095_half_map_2.map.gz emd_38095_half_map_2.map.gz | 117.8 MB 115.9 MB 115.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38095 http://ftp.pdbj.org/pub/emdb/structures/EMD-38095 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38095 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38095 | HTTPS FTP |
-Validation report
| Summary document |  emd_38095_validation.pdf.gz emd_38095_validation.pdf.gz | 1.5 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_38095_full_validation.pdf.gz emd_38095_full_validation.pdf.gz | 1.5 MB | Display | |
| Data in XML |  emd_38095_validation.xml.gz emd_38095_validation.xml.gz | 13.7 KB | Display | |
| Data in CIF |  emd_38095_validation.cif.gz emd_38095_validation.cif.gz | 16.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38095 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38095 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38095 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38095 | HTTPS FTP |
-Related structure data
| Related structure data |  8x79MC  8x7aC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_38095.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38095.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.73 Å | ||||||||||||||||||||||||||||||||||||
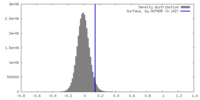
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_38095_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
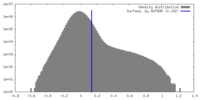
| Density Histograms |
-Half map: #2
| File | emd_38095_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
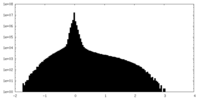
| Density Histograms |
-Half map: #1
| File | emd_38095_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
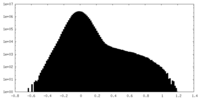
| Density Histograms |
- Sample components
Sample components
-Entire : IP-Gs complex
| Entire | Name: IP-Gs complex |
|---|---|
| Components |
|
-Supramolecule #1: IP-Gs complex
| Supramolecule | Name: IP-Gs complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #5, #1-#4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms sh...
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short,GNAS complex locus,GNAS complex locus type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 29.068906 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MNSKTEDQRN EEKAQREANK KIEKQLQKDK QVYRATHRLL LLGADNSGKS TIVKQMRILH GGSGGSGGTS GIFETKFQVD KVNFHMFDV GAQRDERRKW IQCFNDVTAI IFVVDSSDYN RLQEALNLFK SIWNNRWLRT ISVILFLNKQ DLLAEKVLAG K SKIEDYFP ...String: MNSKTEDQRN EEKAQREANK KIEKQLQKDK QVYRATHRLL LLGADNSGKS TIVKQMRILH GGSGGSGGTS GIFETKFQVD KVNFHMFDV GAQRDERRKW IQCFNDVTAI IFVVDSSDYN RLQEALNLFK SIWNNRWLRT ISVILFLNKQ DLLAEKVLAG K SKIEDYFP EFARYTTPED ATPEPGEDPR VTRAKYFIRD EFLRISTASG DGRHYCYPHF TCSVDTENAR RIFNDCRDII QR MHLRQYE LL UniProtKB: Guanine nucleotide-binding protein G(s) subunit alpha |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.613176 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: HHHHHHGSLL QSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV ...String: HHHHHHGSLL QSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV TSSGDTTCAL WDIETGQQTT TFTGHTGDVM SLSLAPDTRL FVSGACDASA KLWDVREGMC RQTFTGHESD IN AICFFPN GNAFATGSDD ATCRLFDLRA DQELMTYSHD NIICGITSVS FSKSGRLLLA GYDDFNCNVW DALKADRAGV LAG HDNRVS CLGVTDDGMA VATGSWDSFL KIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: Nanobody35
| Macromolecule | Name: Nanobody35 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 14.71432 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTVSSH HHHHH |
-Macromolecule #5: Prostacyclin receptor
| Macromolecule | Name: Prostacyclin receptor / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.989914 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MADSCRNLTY VRGSVGPATS TLMFVAGVVG NGLALGILSA RRPARPSAFA VLVTGLAATD LLGTSFLSPA VFVAYARNSS LLGLARGGP ALCDAFAFAM TFFGLASMLI LFAMAVERCL ALSHPYLYAQ LDGPRCARLA LPAIYAFCVL FCALPLLGLG Q HQQYCPGS ...String: MADSCRNLTY VRGSVGPATS TLMFVAGVVG NGLALGILSA RRPARPSAFA VLVTGLAATD LLGTSFLSPA VFVAYARNSS LLGLARGGP ALCDAFAFAM TFFGLASMLI LFAMAVERCL ALSHPYLYAQ LDGPRCARLA LPAIYAFCVL FCALPLLGLG Q HQQYCPGS WCFLRMRWAQ PGGAAFSLAY AGLVALLVAA IFLCNGSVTL SLCRMYRQQK RHQGSLGPRP RTGEDEVDHL IL LALMTVV MAVCSLPLTI RCFTQAVAPD SSSEMGDLLA FRFYAFNPIL DPWVFILFRK AVFQRLKLWV CCLCLGPAHG DSQ TPLSQL ASGRRDPRAP SAPVGKEGSC VPLSAWGEGQ VEPLPPTQQS SGSAVGTSSK AEASVACSLC UniProtKB: Prostacyclin receptor |
-Macromolecule #6: 2-[4-[(5,6-diphenylpyrazin-2-yl)-propan-2-yl-amino]butoxy]ethanoi...
| Macromolecule | Name: 2-[4-[(5,6-diphenylpyrazin-2-yl)-propan-2-yl-amino]butoxy]ethanoic acid type: ligand / ID: 6 / Number of copies: 1 / Formula: Y9Q |
|---|---|
| Molecular weight | Theoretical: 419.516 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 5.0 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.41 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 206075 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
-Atomic model buiding 1
| Refinement | Protocol: OTHER |
|---|---|
| Output model |  PDB-8x79: |
 Movie
Movie Controller
Controller



























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)