+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | SIRM reconstruction of the unpublished protein | |||||||||
 Map data Map data | SIRM reconstruction of the unpublished protein, after Validation and Mask-Picking. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Unpublished protein / MEMBRANE PROTEIN | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Zhang XZ / Zhu DJ / Cao WL | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2024 Journal: Sci Adv / Year: 2024Title: Correction of preferred orientation-induced distortion in cryo-electron microscopy maps. Authors: Dongjie Zhu / Weili Cao / Junxi Li / Chunling Wu / Duanfang Cao / Xinzheng Zhang /  Abstract: Reconstruction maps of cryo-electron microscopy (cryo-EM) exhibit distortion when the cryo-EM dataset is incomplete, usually caused by unevenly distributed orientations. Prior efforts had been ...Reconstruction maps of cryo-electron microscopy (cryo-EM) exhibit distortion when the cryo-EM dataset is incomplete, usually caused by unevenly distributed orientations. Prior efforts had been attempted to address this preferred orientation problem using tilt-collection strategy and modifications to grids or to air-water interfaces. However, these approaches often require time-consuming experiments, and the effect was always protein dependent. Here, we developed a procedure containing removing misaligned particles and an iterative reconstruction method based on signal-to-noise ratio of Fourier component to correct this distortion by recovering missing data using a purely computational algorithm. This procedure called signal-to-noise ratio iterative reconstruction method (SIRM) was applied on incomplete datasets of various proteins to fix distortion in cryo-EM maps and to a more isotropic resolution. In addition, SIRM provides a better reference map for further reconstruction refinements, resulting in an improved alignment, which ultimately improves map quality and benefits model building. | |||||||||
| History |
|
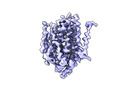
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38082.map.gz emd_38082.map.gz | 27.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38082-v30.xml emd-38082-v30.xml emd-38082.xml emd-38082.xml | 12.8 KB 12.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_38082.png emd_38082.png | 49.4 KB | ||
| Masks |  emd_38082_msk_1.map emd_38082_msk_1.map | 30.5 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-38082.cif.gz emd-38082.cif.gz | 3.8 KB | ||
| Others |  emd_38082_half_map_1.map.gz emd_38082_half_map_1.map.gz emd_38082_half_map_2.map.gz emd_38082_half_map_2.map.gz | 23.4 MB 23.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38082 http://ftp.pdbj.org/pub/emdb/structures/EMD-38082 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38082 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38082 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_38082.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38082.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | SIRM reconstruction of the unpublished protein, after Validation and Mask-Picking. | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.167 Å | ||||||||||||||||||||||||||||||||||||
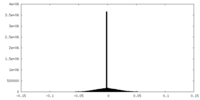
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_38082_msk_1.map emd_38082_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
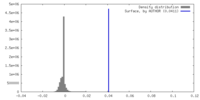
| Density Histograms |
-Half map: Conventional reconstruction of the unpublished protein, after Validation...
| File | emd_38082_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Conventional reconstruction of the unpublished protein, after Validation and Mask-Picking. Only used for measuring FSC. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Conventional reconstruction of the unpublished protein, after Validation...
| File | emd_38082_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Conventional reconstruction of the unpublished protein, after Validation and Mask-Picking. Only used for measuring FSC. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Unpublished protein
| Entire | Name: Unpublished protein |
|---|---|
| Components |
|
-Supramolecule #1: Unpublished protein
| Supramolecule | Name: Unpublished protein / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 52.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.8 Å / Resolution method: OTHER / Software - Name: RELION (ver. 3.1.2) / Software - details: SIRM-RELION Details: The main map was low-passed to 3.8 Angstrom. The reported resolution via FSC-0.143 Gold-standard was 3.3 Angstrom Number images used: 74350 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.1.2) |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)