[English] 日本語
 Yorodumi
Yorodumi- EMDB-37408: De novo transcribing complex 14 (TC14), the early elongation comp... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | De novo transcribing complex 14 (TC14), the early elongation complex with Pol II positioned 14nt downstream of TSS | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | transcribing complex / de novo transcription initiation / early elongation complex (EEC) / TRANSCRIPTION | |||||||||
| Function / homology |  Function and homology information Function and homology informationphosphatase activator activity / TFIIF-class transcription factor complex binding / transcription factor TFIIF complex / Formation of RNA Pol II elongation complex / Formation of the Early Elongation Complex / Transcriptional regulation by small RNAs / RNA Polymerase II Pre-transcription Events / TP53 Regulates Transcription of DNA Repair Genes / FGFR2 alternative splicing / RNA polymerase II transcribes snRNA genes ...phosphatase activator activity / TFIIF-class transcription factor complex binding / transcription factor TFIIF complex / Formation of RNA Pol II elongation complex / Formation of the Early Elongation Complex / Transcriptional regulation by small RNAs / RNA Polymerase II Pre-transcription Events / TP53 Regulates Transcription of DNA Repair Genes / FGFR2 alternative splicing / RNA polymerase II transcribes snRNA genes / mRNA Capping / mRNA Splicing - Minor Pathway / Processing of Capped Intron-Containing Pre-mRNA / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Elongation / RNA Polymerase II Transcription Initiation And Promoter Clearance / RNA Pol II CTD phosphorylation and interaction with CE / Estrogen-dependent gene expression / Formation of TC-NER Pre-Incision Complex / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / mRNA Splicing - Major Pathway / RNA polymerase II general transcription initiation factor binding / Abortive elongation of HIV-1 transcript in the absence of Tat / FGFR2 alternative splicing / Viral Messenger RNA Synthesis / Signaling by FGFR2 IIIa TM / transcription factor TFIID complex / RNA polymerase II general transcription initiation factor activity / RNA Pol II CTD phosphorylation and interaction with CE during HIV infection / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / HIV Transcription Initiation / RNA Polymerase II HIV Promoter Escape / Transcription of the HIV genome / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Initiation And Promoter Clearance / mRNA Splicing - Minor Pathway / Processing of Capped Intron-Containing Pre-mRNA / maintenance of transcriptional fidelity during transcription elongation by RNA polymerase II / RNA polymerase II transcribes snRNA genes / Pausing and recovery of Tat-mediated HIV elongation / Tat-mediated HIV elongation arrest and recovery / HIV elongation arrest and recovery / Pausing and recovery of HIV elongation / negative regulation of protein binding / Tat-mediated elongation of the HIV-1 transcript / RNA polymerase I complex / Formation of HIV-1 elongation complex containing HIV-1 Tat / RNA polymerase III complex / Formation of HIV elongation complex in the absence of HIV Tat / RNA polymerase II, core complex / tRNA transcription by RNA polymerase III / transcription by RNA polymerase I / RNA Polymerase II Transcription Elongation / Formation of RNA Pol II elongation complex / transcription-coupled nucleotide-excision repair / RNA Polymerase II Pre-transcription Events / mRNA Splicing - Major Pathway / DNA-directed RNA polymerase complex / TP53 Regulates Transcription of DNA Repair Genes / transcription initiation at RNA polymerase II promoter / DNA-templated transcription initiation / promoter-specific chromatin binding / positive regulation of transcription elongation by RNA polymerase II / transcription elongation by RNA polymerase II / ribonucleoside binding / fibrillar center / response to virus / DNA-directed RNA polymerase / DNA-directed RNA polymerase activity / cell junction / chromosome / toxin activity / microtubule cytoskeleton / protein phosphatase binding / transcription by RNA polymerase II / Estrogen-dependent gene expression / nucleic acid binding / protein dimerization activity / protein domain specific binding / nucleotide binding / hydrolase activity / RNA-directed RNA polymerase activity / DNA-templated transcription / nucleolus / positive regulation of transcription by RNA polymerase II / protein-containing complex / DNA binding / RNA binding / zinc ion binding / nucleoplasm / metal ion binding / nucleus Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / synthetic construct (others) / Homo sapiens (human) / synthetic construct (others) /  Amanita phalloides (death cap) Amanita phalloides (death cap) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.75 Å | |||||||||
 Authors Authors | Chen X / Liu W / Wang Q / Wang X / Ren Y / Qu X / Li W / Xu Y | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2023 Journal: Science / Year: 2023Title: Structural visualization of transcription initiation in action. Authors: Xizi Chen / Weida Liu / Qianmin Wang / Xinxin Wang / Yulei Ren / Xuechun Qu / Wanjun Li / Yanhui Xu /  Abstract: Transcription initiation is a complex process, and its mechanism is incompletely understood. We determined the structures of de novo transcribing complexes TC2 to TC17 with RNA polymerase II halted ...Transcription initiation is a complex process, and its mechanism is incompletely understood. We determined the structures of de novo transcribing complexes TC2 to TC17 with RNA polymerase II halted on G-less promoters when nascent RNAs reach 2 to 17 nucleotides in length, respectively. Connecting these structures generated a movie and a working model. As initially synthesized RNA grows, general transcription factors (GTFs) remain bound to the promoter and the transcription bubble expands. Nucleoside triphosphate (NTP)-driven RNA-DNA translocation and template-strand accumulation in a nearly sealed channel may promote the transition from initially transcribing complexes (ITCs) (TC2 to TC9) to early elongation complexes (EECs) (TC10 to TC17). Our study shows dynamic processes of transcription initiation and reveals why ITCs require GTFs and bubble expansion for initial RNA synthesis, whereas EECs need GTF dissociation from the promoter and bubble collapse for promoter escape. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37408.map.gz emd_37408.map.gz | 2.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37408-v30.xml emd-37408-v30.xml emd-37408.xml emd-37408.xml | 47.9 KB 47.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_37408.png emd_37408.png | 135.1 KB | ||
| Filedesc metadata |  emd-37408.cif.gz emd-37408.cif.gz | 10.9 KB | ||
| Others |  emd_37408_additional_1.map.gz emd_37408_additional_1.map.gz emd_37408_half_map_1.map.gz emd_37408_half_map_1.map.gz emd_37408_half_map_2.map.gz emd_37408_half_map_2.map.gz | 74.6 MB 70.1 MB 70.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37408 http://ftp.pdbj.org/pub/emdb/structures/EMD-37408 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37408 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37408 | HTTPS FTP |
-Related structure data
| Related structure data |  8waxMC  8wakC  8walC  8wanC  8waoC  8wapC  8waqC  8warC  8wasC  8watC  8wauC  8wavC  8wawC  8wayC  8wazC  8wb0C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_37408.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37408.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.334 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Overall map of TC14
| File | emd_37408_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Overall map of TC14 | ||||||||||||
| Projections & Slices |
| ||||||||||||
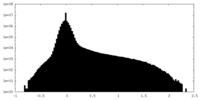
| Density Histograms |
-Half map: #1
| File | emd_37408_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
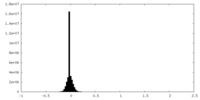
| Density Histograms |
-Half map: #2
| File | emd_37408_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
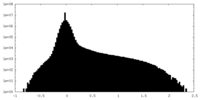
| Density Histograms |
- Sample components
Sample components
+Entire : Structure of transcribing complex 14 (TC14), the early elongation...
+Supramolecule #1: Structure of transcribing complex 14 (TC14), the early elongation...
+Supramolecule #2: RNA POLYMERASE II
+Supramolecule #3: TFIIF
+Supramolecule #4: DNA
+Supramolecule #5: RNA
+Supramolecule #6: Alpha-amanitin
+Macromolecule #1: Alpha-amanitin
+Macromolecule #2: General transcription factor IIF subunit 1
+Macromolecule #3: General transcription factor IIF subunit 2
+Macromolecule #7: DNA-directed RNA polymerase subunit
+Macromolecule #8: DNA-directed RNA polymerase subunit beta
+Macromolecule #9: DNA-directed RNA polymerase II subunit RPB3
+Macromolecule #10: DNA-directed RNA polymerase II subunit RPB4
+Macromolecule #11: DNA-directed RNA polymerase II subunit E
+Macromolecule #12: DNA-directed RNA polymerase II subunit F
+Macromolecule #13: DNA-directed RNA polymerase II subunit RPB7
+Macromolecule #14: DNA-directed RNA polymerases I, II, and III subunit RPABC3
+Macromolecule #15: DNA-directed RNA polymerase II subunit RPB9
+Macromolecule #16: DNA-directed RNA polymerases I, II, and III subunit RPABC5
+Macromolecule #17: DNA-directed RNA polymerase II subunit RPB11-a
+Macromolecule #18: RPB12
+Macromolecule #4: non-template DNA
+Macromolecule #5: template DNA
+Macromolecule #6: RNA
+Macromolecule #19: ZINC ION
+Macromolecule #20: MAGNESIUM ION
+Macromolecule #21: [[(2~{S},3~{R},4~{S},5~{R})-5-(2-azanyl-6-oxidanylidene-3~{H}-pur...
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.9 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.75 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 261023 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller







































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)