[English] 日本語
 Yorodumi
Yorodumi- EMDB-34724: Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis s... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis sp. PCC 6803 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CpcL-phycobilisome / energy transfer / ferredoxin:NADP+ oxidoreductase / ultrafast spectroscopy / PHOTOSYNTHESIS | |||||||||
| Function / homology |  Function and homology information Function and homology informationferredoxin-NADP+ reductase / ferredoxin-NADP+ reductase activity / phycobilisome / plasma membrane-derived thylakoid membrane / photosynthesis Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.64 Å | |||||||||
 Authors Authors | Zheng L / Zhang Z / Wang H / Zheng Z / Gao N / Zhao J | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Cryo-EM and femtosecond spectroscopic studies provide mechanistic insight into the energy transfer in CpcL-phycobilisomes. Authors: Lvqin Zheng / Zhengdong Zhang / Hongrui Wang / Zhenggao Zheng / Jiayu Wang / Heyuan Liu / Hailong Chen / Chunxia Dong / Guopeng Wang / Yuxiang Weng / Ning Gao / Jindong Zhao /  Abstract: Phycobilisomes (PBS) are the major light harvesting complexes of photosynthesis in the cyanobacteria and red algae. CpcL-PBS is a type of small PBS in cyanobacteria that transfers energy directly to ...Phycobilisomes (PBS) are the major light harvesting complexes of photosynthesis in the cyanobacteria and red algae. CpcL-PBS is a type of small PBS in cyanobacteria that transfers energy directly to photosystem I without the core structure. Here we report the cryo-EM structure of the CpcL-PBS from the cyanobacterium Synechocystis sp. PCC 6803 at 2.6-Å resolution. The structure shows the CpcD domain of ferredoxin: NADP oxidoreductase is located at the distal end of CpcL-PBS, responsible for its attachment to PBS. With the evidence of ultrafast transient absorption and fluorescence spectroscopy, the roles of individual bilins in energy transfer are revealed. The bilin β located near photosystem I has an enhanced planarity and is the red-bilin responsible for the direct energy transfer to photosystem I. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34724.map.gz emd_34724.map.gz | 16.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34724-v30.xml emd-34724-v30.xml emd-34724.xml emd-34724.xml | 24.3 KB 24.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34724.png emd_34724.png | 37.6 KB | ||
| Filedesc metadata |  emd-34724.cif.gz emd-34724.cif.gz | 7.1 KB | ||
| Others |  emd_34724_half_map_1.map.gz emd_34724_half_map_1.map.gz emd_34724_half_map_2.map.gz emd_34724_half_map_2.map.gz | 140.9 MB 140.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34724 http://ftp.pdbj.org/pub/emdb/structures/EMD-34724 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34724 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34724 | HTTPS FTP |
-Related structure data
| Related structure data |  8hfqMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34724.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34724.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34724_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_34724_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis s...
| Entire | Name: Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis sp. PCC 6803 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis s...
| Supramolecule | Name: Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis sp. PCC 6803 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: C-phycocyanin alpha subunit
| Macromolecule | Name: C-phycocyanin alpha subunit / type: protein_or_peptide / ID: 1 / Number of copies: 18 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 17.602529 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTPLTEAVS TADSQGRFLS STELQIAFGR LRQANAGLQA AKALTDNAQS LVNGAAQAVY NKFPYTTQTQ GNNFAADQRG KDKCARDIG YYLRIVTYCL VAGGTGPLDE YLIAGIDEIN RTFDLSPSWY VEALKYIKAN HGLSGDARDE ANSYLDYAIN A LS UniProtKB: C-phycocyanin alpha subunit |
-Macromolecule #2: C-phycocyanin beta subunit
| Macromolecule | Name: C-phycocyanin beta subunit / type: protein_or_peptide / ID: 2 / Number of copies: 18 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 18.142426 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MFDVFTRVVS QADARGEYLS GSQLDALSAT VAEGNKRIDS VNRITGNASA IVSNAARALF AEQPQLIQPG GNAYTSRRMA ACLRDMEII LRYVTYATFT GDASVLEDRC LNGLRETYVA LGVPGASVAA GVQKMKEAAL DIVNDPNGIT RGDCSAIVAE I AGYFDRAA AAVA UniProtKB: C-phycocyanin beta subunit |
-Macromolecule #3: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated...
| Macromolecule | Name: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 32.558607 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAITTAASRL GVAPYNESRP VELRPDFSLD DAKMVIRAVY RQVLGNDYIM DSERLKGAES LLTNGSISVR EFVRTVAKSE LYKKKFLYN NFQTRVIELN YKHLLGRAPF SEDEVIFHLD LYENQGFDAD IDSYIDSVEY QENFGENIVP YYRFNNQVGD R TVGFTRMF ...String: MAITTAASRL GVAPYNESRP VELRPDFSLD DAKMVIRAVY RQVLGNDYIM DSERLKGAES LLTNGSISVR EFVRTVAKSE LYKKKFLYN NFQTRVIELN YKHLLGRAPF SEDEVIFHLD LYENQGFDAD IDSYIDSVEY QENFGENIVP YYRFNNQVGD R TVGFTRMF RLYRGYANSD RSQLERSSSR LATELGQNTV SAIVGPSGSN AGWAYRPSRA GNTPAKALGG TVPFGQASKL FR VEITAIS APGYPKVRRS NKAVIVPFEQ LNQTLQQINR LGGKVASITP ASLS UniProtKB: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 1 |
-Macromolecule #4: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated...
| Macromolecule | Name: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 30.836346 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTSLVSAQRL GIVAVDEAIP LELRSRSTEE EVDAVILAVY RQVLGNDHLM SQERLTSAES LLRGREISVR DFVRAVALSE VYRQKFFHS NPQNRFIELN YKHLLGRAPY DQSEIAFHTD LYHQGGYEAE INSYIDSVEY TENFGDWVVP YFRGFATQRN Q KTVGFSRS ...String: MTSLVSAQRL GIVAVDEAIP LELRSRSTEE EVDAVILAVY RQVLGNDHLM SQERLTSAES LLRGREISVR DFVRAVALSE VYRQKFFHS NPQNRFIELN YKHLLGRAPY DQSEIAFHTD LYHQGGYEAE INSYIDSVEY TENFGDWVVP YFRGFATQRN Q KTVGFSRS FQVYRGYATS DRSQGNGSRS RLTRELARNT ASPVYAGSTA ESLRGTSAGS RNQMYRLQVI QGAAPGRGTR VR RGKAEYL VSYDNLSAKL QQINRQGDTV TMISLA UniProtKB: Phycobilisome 32.1 kDa linker polypeptide, phycocyanin-associated, rod 2 |
-Macromolecule #5: Ferredoxin--NADP reductase
| Macromolecule | Name: Ferredoxin--NADP reductase / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO / EC number: ferredoxin-NADP+ reductase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 46.417469 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MYSPGYVATS SRQSDAGNRL FVYEVIGLSQ STMTDGLDYP IRRSGSTFIT VPLKRMNQEM RRITRMGGKI VSIKPLEGDS PLPHTEGIA KPSQSEGSGS EAVANPAPES NKTMTTTPKE KKADDIPVNI YRPKTPYIGK VLENYPLVRE GAIGTVQHLT F DLSAGDLR ...String: MYSPGYVATS SRQSDAGNRL FVYEVIGLSQ STMTDGLDYP IRRSGSTFIT VPLKRMNQEM RRITRMGGKI VSIKPLEGDS PLPHTEGIA KPSQSEGSGS EAVANPAPES NKTMTTTPKE KKADDIPVNI YRPKTPYIGK VLENYPLVRE GAIGTVQHLT F DLSAGDLR YLEGQSIGII PPGEDDKGKP HKLRLYSIAS TRHGDFGDDK TVSLCVRQLE YQNEAGETVQ GVCSTYLCNI KE GDDIAIT GPVGKEMLLP PDEDANIVML ATGTGIAPFR AFLWRMFKEQ HEDYKFKGLA WLIFGIPKSE NILYKDDLEK MAA EFPDNF RLTYAISREQ QNAEGGRMYI QHRVAENAEE LWNLMQNPKT HTYMCGLKGM EPGIDEAFTA LAEQNGKEWT TFQR EMKKE HRWHVETY UniProtKB: Ferredoxin--NADP reductase |
-Macromolecule #6: Photosystem I-associated linker protein CpcL
| Macromolecule | Name: Photosystem I-associated linker protein CpcL / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 28.551516 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTLPLIAYAP VSQNQRVTNY EVSGDEHARI FTTEGTLSPS AMDNLIAAAY RQVFNEQQMI QSNRQIALES QFKNQQITVR DFIRGLALS DSFRRRNFEV NNNYRFVQMC IQRLLGRDVY SEEEKIAWSI VIATKGLPGF INELLNSQEY LENFGYDTVP Y QRRRILPQ ...String: MTLPLIAYAP VSQNQRVTNY EVSGDEHARI FTTEGTLSPS AMDNLIAAAY RQVFNEQQMI QSNRQIALES QFKNQQITVR DFIRGLALS DSFRRRNFEV NNNYRFVQMC IQRLLGRDVY SEEEKIAWSI VIATKGLPGF INELLNSQEY LENFGYDTVP Y QRRRILPQ RISGELPFAR MPRYGADHRE KLEAIGYFRN QAPLTYRWEW QKQPYPAGVY LAGKVVLYVG GALVSLGIIA VA LSAWGII GL UniProtKB: Photosystem I-associated linker protein CpcL |
-Macromolecule #7: PHYCOCYANOBILIN
| Macromolecule | Name: PHYCOCYANOBILIN / type: ligand / ID: 7 / Number of copies: 54 / Formula: CYC |
|---|---|
| Molecular weight | Theoretical: 588.694 Da |
| Chemical component information | 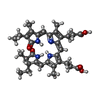 ChemComp-CYC: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
| Details | Cryo-EM structure of CpcL-PBS from cyanobacterium Synechocystis sp. PCC 6803 |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)