+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human ATP synthase F1 domain, state2 | |||||||||||||||
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / Cristae formation / estradiol binding / angiostatin binding / ATP biosynthetic process / Mitochondrial protein import / oxidative phosphorylation / cellular response to interleukin-7 / response to muscle activity ...negative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / Cristae formation / estradiol binding / angiostatin binding / ATP biosynthetic process / Mitochondrial protein import / oxidative phosphorylation / cellular response to interleukin-7 / response to muscle activity / negative regulation of endothelial cell proliferation / cellular response to cytokine stimulus / proton motive force-driven ATP synthesis / proton motive force-driven mitochondrial ATP synthesis / mitochondrial nucleoid / proton-transporting ATPase activity, rotational mechanism / MHC class I protein binding / positive regulation of blood vessel endothelial cell migration / cellular response to dexamethasone stimulus / H+-transporting two-sector ATPase / proton-transporting ATP synthase complex / proton-transporting ATP synthase activity, rotational mechanism / cellular response to nitric oxide / Mitochondrial protein degradation / proton transmembrane transport / cellular response to cAMP / regulation of intracellular pH / lipid metabolic process / generation of precursor metabolites and energy / sperm end piece / Transcriptional activation of mitochondrial biogenesis / ADP binding / mitochondrial membrane / osteoblast differentiation / protease binding / angiogenesis / response to ethanol / mitochondrial inner membrane / membrane raft / mitochondrial matrix / protein-containing complex binding / cell surface / ATP hydrolysis activity / mitochondrion / RNA binding / extracellular exosome / ATP binding / membrane / metal ion binding / nucleus / plasma membrane Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.77 Å | |||||||||||||||
 Authors Authors | Lai Y / Zhang Y / Liu F / Gao Y / Gong H / Rao Z | |||||||||||||||
| Funding support |  China, 4 items China, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Structure of the human ATP synthase. Authors: Yuezheng Lai / Yuying Zhang / Shan Zhou / Jinxu Xu / Zhanqiang Du / Ziyan Feng / Long Yu / Ziqing Zhao / Weiwei Wang / Yanting Tang / Xiuna Yang / Luke W Guddat / Fengjiang Liu / Yan Gao / ...Authors: Yuezheng Lai / Yuying Zhang / Shan Zhou / Jinxu Xu / Zhanqiang Du / Ziyan Feng / Long Yu / Ziqing Zhao / Weiwei Wang / Yanting Tang / Xiuna Yang / Luke W Guddat / Fengjiang Liu / Yan Gao / Zihe Rao / Hongri Gong /   Abstract: Biological energy currency ATP is produced by FF-ATP synthase. However, the molecular mechanism for human ATP synthase action remains unknown. Here, we present snapshot images for three main ...Biological energy currency ATP is produced by FF-ATP synthase. However, the molecular mechanism for human ATP synthase action remains unknown. Here, we present snapshot images for three main rotational states and one substate of human ATP synthase using cryoelectron microscopy. These structures reveal that the release of ADP occurs when the β subunit of FF-ATP synthase is in the open conformation, showing how ADP binding is coordinated during synthesis. The accommodation of the symmetry mismatch between F and F motors is resolved by the torsional flexing of the entire complex, especially the γ subunit, and the rotational substep of the c subunit. Water molecules are identified in the inlet and outlet half-channels, suggesting that the proton transfer in these two half-channels proceed via a Grotthus mechanism. Clinically relevant mutations are mapped to the structure, showing that they are mainly located at the subunit-subunit interfaces, thus causing instability of the complex. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34568.map.gz emd_34568.map.gz | 484 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34568-v30.xml emd-34568-v30.xml emd-34568.xml emd-34568.xml | 24 KB 24 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34568.png emd_34568.png | 151.8 KB | ||
| Filedesc metadata |  emd-34568.cif.gz emd-34568.cif.gz | 7.1 KB | ||
| Others |  emd_34568_half_map_1.map.gz emd_34568_half_map_1.map.gz emd_34568_half_map_2.map.gz emd_34568_half_map_2.map.gz | 474.4 MB 474.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34568 http://ftp.pdbj.org/pub/emdb/structures/EMD-34568 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34568 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34568 | HTTPS FTP |
-Related structure data
| Related structure data |  8h9iMC  8h9eC  8h9fC  8h9gC  8h9jC  8h9kC  8h9lC  8h9mC  8h9nC  8h9pC  8h9qC  8h9rC  8h9sC  8h9tC  8h9uC  8h9vC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34568.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34568.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.73 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34568_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
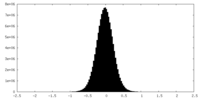
| Density Histograms |
-Half map: #1
| File | emd_34568_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
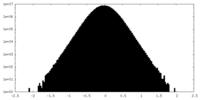
| Density Histograms |
- Sample components
Sample components
-Entire : Human ATP synthase
| Entire | Name: Human ATP synthase |
|---|---|
| Components |
|
-Supramolecule #1: Human ATP synthase
| Supramolecule | Name: Human ATP synthase / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1, #3-#4, #2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 600 KDa |
-Macromolecule #1: ATP synthase subunit alpha, mitochondrial
| Macromolecule | Name: ATP synthase subunit alpha, mitochondrial / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 55.27616 KDa |
| Sequence | String: QKTGTAEMSS ILEERILGAD TSVDLEETGR VLSIGDGIAR VHGLRNVQAE EMVEFSSGLK GMSLNLEPDN VGVVVFGNDK LIKEGDIVK RTGAIVDVPV GEELLGRVVD ALGNAIDGKG PIGSKTRRRV GLKAPGIIPR ISVREPMQTG IKAVDSLVPI G RGQRELII ...String: QKTGTAEMSS ILEERILGAD TSVDLEETGR VLSIGDGIAR VHGLRNVQAE EMVEFSSGLK GMSLNLEPDN VGVVVFGNDK LIKEGDIVK RTGAIVDVPV GEELLGRVVD ALGNAIDGKG PIGSKTRRRV GLKAPGIIPR ISVREPMQTG IKAVDSLVPI G RGQRELII GDRQTGKTSI AIDTIINQKR FNDGSDEKKK LYCIYVAIGQ KRSTVAQLVK RLTDADAMKY TIVVSATASD AA PLQYLAP YSGCSMGEYF RDNGKHALII YDDLSKQAVA YRQMSLLLRR PPGREAYPGD VFYLHSRLLE RAAKMNDAFG GGS LTALPV IETQAGDVSA YIPTNVISIT DGQIFLETEL FYKGIRPAIN VGLSVSRVGS AAQTRAMKQV AGTMKLELAQ YREV AAFAQ FGSDLDAATQ QLLSRGVRLT ELLKQGQYSP MAIEEQVAVI YAGVRGYLDK LEPSKITKFE NAFLSHVVSQ HQALL GTIR ADGKISEQSD AKLKEIVTNF LAGFEA UniProtKB: ATP synthase F(1) complex subunit alpha, mitochondrial |
-Macromolecule #2: ATP synthase subunit beta, mitochondrial
| Macromolecule | Name: ATP synthase subunit beta, mitochondrial / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO / EC number: H+-transporting two-sector ATPase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 51.821965 KDa |
| Sequence | String: AQTSPSPKAG AATGRIVAVI GAVVDVQFDE GLPPILNALE VQGRETRLVL EVAQHLGEST VRTIAMDGTE GLVRGQKVLD SGAPIKIPV GPETLGRIMN VIGEPIDERG PIKTKQFAPI HAEAPEFMEM SVEQEILVTG IKVVDLLAPY AKGGKIGLFG G AGVGKTVL ...String: AQTSPSPKAG AATGRIVAVI GAVVDVQFDE GLPPILNALE VQGRETRLVL EVAQHLGEST VRTIAMDGTE GLVRGQKVLD SGAPIKIPV GPETLGRIMN VIGEPIDERG PIKTKQFAPI HAEAPEFMEM SVEQEILVTG IKVVDLLAPY AKGGKIGLFG G AGVGKTVL IMELINNVAK AHGGYSVFAG VGERTREGND LYHEMIESGV INLKDATSKV ALVYGQMNEP PGARARVALT GL TVAEYFR DQEGQDVLLF IDNIFRFTQA GSEVSALLGR IPSAVGYQPT LATDMGTMQE RITTTKKGSI TSVQAIYVPA DDL TDPAPA TTFAHLDATT VLSRAIAELG IYPAVDPLDS TSRIMDPNIV GSEHYDVARG VQKILQDYKS LQDIIAILGM DELS EEDKL TVSRARKIQR FLSQPFQVAE VFTGHMGKLV PLKETIKGFQ QILAGEYDHL PEQAFYMVGP IEEAVAKADK LAEEH SS UniProtKB: ATP synthase F(1) complex subunit beta, mitochondrial |
-Macromolecule #3: ATP synthase subunit gamma, mitochondrial
| Macromolecule | Name: ATP synthase subunit gamma, mitochondrial / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 30.207752 KDa |
| Sequence | String: ATLKDITRRL KSIKNIQKIT KSMKMVAAAK YARAERELKP ARIYGLGSLA LYEKADIKGP EDKKKHLLIG VSSDRGLCGA IHSSIAKQM KSEVATLTAA GKEVMLVGIG DKIRGILYRT HSDQFLVAFK EVGRKPPTFG DASVIALELL NSGYEFDEGS I IFNKFRSV ...String: ATLKDITRRL KSIKNIQKIT KSMKMVAAAK YARAERELKP ARIYGLGSLA LYEKADIKGP EDKKKHLLIG VSSDRGLCGA IHSSIAKQM KSEVATLTAA GKEVMLVGIG DKIRGILYRT HSDQFLVAFK EVGRKPPTFG DASVIALELL NSGYEFDEGS I IFNKFRSV ISYKTEEKPI FSLNTVASAD SMSIYDDIDA DVLQNYQEYN LANIIYYSLK ESTTSEQSAR MTAMDNASKN AS EMIDKLT LTFNRTRQAV ITKELIEIIS GAAALD UniProtKB: ATP synthase F(1) complex subunit gamma, mitochondrial |
-Macromolecule #4: ATP synthase subunit O, mitochondrial
| Macromolecule | Name: ATP synthase subunit O, mitochondrial / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 20.904488 KDa |
| Sequence | String: FAKLVRPPVQ VYGIEGRYAT ALYSAASKQN KLEQVEKELL RVAQILKEPK VAASVLNPYV KRSIKVKSLN DITAKERFSP LTTNLINLL AENGRLSNTQ GVVSAFSTMM SVHRGEVPCT VTSASPLEEA TLSELKTVLK SFLSQGQVLK LEAKTDPSIL G GMIVRIGE KYVDMSVKTK IQKLGRAMRE IV UniProtKB: ATP synthase peripheral stalk subunit OSCP, mitochondrial |
-Macromolecule #5: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 5 / Number of copies: 3 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 5 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #7: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 7 / Number of copies: 2 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: TFS Selectris X / Energy filter - Slit width: 10 eV |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.2 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)