+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
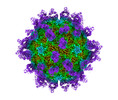
| タイトル | Cryo-EM structure of Coxsackievirus B1 pre-A particle in complex with nAb 2E6 (CVB1-pre-A:2E6) | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Coxsackievirus B1 / Neutralizing antiboy / Cryo-EM / VIRUS | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / picornain 2A / symbiont-mediated suppression of host mRNA export from nucleus / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / ribonucleoside triphosphate phosphatase activity / host cell cytoplasmic vesicle membrane / viral capsid / nucleoside-triphosphate phosphatase ...symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / picornain 2A / symbiont-mediated suppression of host mRNA export from nucleus / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / ribonucleoside triphosphate phosphatase activity / host cell cytoplasmic vesicle membrane / viral capsid / nucleoside-triphosphate phosphatase / host cell / channel activity / monoatomic ion transmembrane transport / DNA replication / RNA helicase activity / endocytosis involved in viral entry into host cell / symbiont-mediated suppression of host gene expression / symbiont-mediated activation of host autophagy / RNA-directed RNA polymerase / cysteine-type endopeptidase activity / viral RNA genome replication / RNA-directed RNA polymerase activity / DNA-templated transcription / symbiont entry into host cell / virion attachment to host cell / host cell nucleus / structural molecule activity / proteolysis / RNA binding / zinc ion binding / ATP binding / membrane 類似検索 - 分子機能 | |||||||||
| 生物種 |    Coxsackievirus B1 (コクサッキーウイルス) Coxsackievirus B1 (コクサッキーウイルス) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.29 Å | |||||||||
 データ登録者 データ登録者 | Zheng Q / Zhu R / Sun H / Cheng T / Li S / Xia N | |||||||||
| 資金援助 | 1件
| |||||||||
 引用 引用 |  ジャーナル: Cell Host Microbe / 年: 2022 ジャーナル: Cell Host Microbe / 年: 2022タイトル: Structural basis for the synergistic neutralization of coxsackievirus B1 by a triple-antibody cocktail. 著者: Qingbing Zheng / Rui Zhu / Zhichao Yin / Longfa Xu / Hui Sun / Hai Yu / Yuanyuan Wu / Yichao Jiang / Qiongzi Huang / Yang Huang / Dongqing Zhang / Liqin Liu / Hongwei Yang / Maozhou He / ...著者: Qingbing Zheng / Rui Zhu / Zhichao Yin / Longfa Xu / Hui Sun / Hai Yu / Yuanyuan Wu / Yichao Jiang / Qiongzi Huang / Yang Huang / Dongqing Zhang / Liqin Liu / Hongwei Yang / Maozhou He / Zhenhong Zhou / Yanan Jiang / Zhenqin Chen / Huan Zhao / Yuqiong Que / Zhibo Kong / Lizhi Zhou / Tingting Li / Jun Zhang / Wenxin Luo / Ying Gu / Tong Cheng / Shaowei Li / Ningshao Xia /  要旨: Coxsackievirus B1 (CVB1) is an emerging pathogen associated with severe neonatal diseases including aseptic meningitis, myocarditis, and pancreatitis and also with the development of type 1 diabetes. ...Coxsackievirus B1 (CVB1) is an emerging pathogen associated with severe neonatal diseases including aseptic meningitis, myocarditis, and pancreatitis and also with the development of type 1 diabetes. We characterize the binding and therapeutic efficacies of three CVB1-specific neutralizing antibodies (nAbs) identified for their ability to inhibit host receptor engagement. High-resolution cryo-EM structures showed that these antibodies recognize different epitopes but with an overlapping region in the capsid VP2 protein and specifically the highly variable EF loop. Moreover, they perturb capsid-receptor interactions by binding various viral particle forms. Antibody combinations achieve synergetic neutralization via a stepwise capsid transition and virion disruption, indicating dynamic changes in the virion in response to multiple nAbs targeting the receptor-binding site. Furthermore, this three-antibody cocktail protects against lethal challenge in neonatal mice and limits pancreatitis and viral replication in a non-obese diabetic mouse model. These results illustrate the utility of nAbs for rational design of therapeutics against picornaviruses such as CVB. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_32968.map.gz emd_32968.map.gz | 632.3 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-32968-v30.xml emd-32968-v30.xml emd-32968.xml emd-32968.xml | 17.6 KB 17.6 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_32968.png emd_32968.png | 145.1 KB | ||
| Filedesc metadata |  emd-32968.cif.gz emd-32968.cif.gz | 6.3 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32968 http://ftp.pdbj.org/pub/emdb/structures/EMD-32968 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32968 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32968 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_32968_validation.pdf.gz emd_32968_validation.pdf.gz | 616.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_32968_full_validation.pdf.gz emd_32968_full_validation.pdf.gz | 616 KB | 表示 | |
| XML形式データ |  emd_32968_validation.xml.gz emd_32968_validation.xml.gz | 8.5 KB | 表示 | |
| CIF形式データ |  emd_32968_validation.cif.gz emd_32968_validation.cif.gz | 9.9 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32968 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32968 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32968 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32968 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7x2iMC  7x2gC  7x2oC  7x2tC  7x2wC  7x35C  7x37C  7x38C  7x3cC  7x3dC  7x3eC  7x3fC  7x3yC  7x40C  7x42C  7x46C  7x47C  7x49C  7x4kC  7x4mC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_32968.map.gz / 形式: CCP4 / 大きさ: 669.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_32968.map.gz / 形式: CCP4 / 大きさ: 669.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.12 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Cryo-EM structure of Coxsackievirus B1 pre-A particle in complex ...
| 全体 | 名称: Cryo-EM structure of Coxsackievirus B1 pre-A particle in complex with nAb 2E6 (CVB1-pre-A:2E6) |
|---|---|
| 要素 |
|
-超分子 #1: Cryo-EM structure of Coxsackievirus B1 pre-A particle in complex ...
| 超分子 | 名称: Cryo-EM structure of Coxsackievirus B1 pre-A particle in complex with nAb 2E6 (CVB1-pre-A:2E6) タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: 2E6 light chain
| 分子 | 名称: 2E6 light chain / タイプ: protein_or_peptide / ID: 1 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 11.72002 KDa |
| 配列 | 文字列: DIQMTQSPAS LSVSVGETVT ITCRASENVY RNLAWYQQKQ GKSPQLLVYA ATNLADGVPS RFSGSGSGTQ YSLKINSLQS EDFGSYFCQ HFWSPVFTFG AGTKLELK |
-分子 #2: 2E6 heavy chain
| 分子 | 名称: 2E6 heavy chain / タイプ: protein_or_peptide / ID: 2 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 13.091568 KDa |
| 配列 | 文字列: QVQLKQSGPG LVQPSQSLSI TCTVSGFSLT NYGVHWVRQS PGKGLEWLGV IWRGGSTDYN AAFMSRLSIT KDNSKSQVFF KMNSLQADD TAIYYCAKGD YYGYDAMDSW GQGTSVTVSR |
-分子 #3: Virion protein 1
| 分子 | 名称: Virion protein 1 / タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Coxsackievirus B1 (コクサッキーウイルス) Coxsackievirus B1 (コクサッキーウイルス) |
| 分子量 | 理論値: 31.207117 KDa |
| 配列 | 文字列: GPVEESVDRA VARVADTISS RPTNSESIPA LTAAETGHTS QVVPSDTMQT RHVKNYHSRS ESSIENFLCR SACVYYATYT NNSKKGFAE WVINTRQVAQ LRRKLELFTY LRFDLELTFV ITSAQQPSTA SSVDAPVQTH QIMYVPPGGP VPTKVKDYAW Q TSTNPSVF ...文字列: GPVEESVDRA VARVADTISS RPTNSESIPA LTAAETGHTS QVVPSDTMQT RHVKNYHSRS ESSIENFLCR SACVYYATYT NNSKKGFAE WVINTRQVAQ LRRKLELFTY LRFDLELTFV ITSAQQPSTA SSVDAPVQTH QIMYVPPGGP VPTKVKDYAW Q TSTNPSVF WTEGNAPPRM SIPFISIGNA YSCFYDGWTQ FSRNGVYGIN TLNNMGTLYM RHVNEAGQGP IKSTVRIYFK PK HVKAWVP RPPRLCQYEK QKNVNFSPIG VTTSRTDIIT T UniProtKB: Genome polyprotein |
-分子 #4: VP2
| 分子 | 名称: VP2 / タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Coxsackievirus B1 (コクサッキーウイルス) Coxsackievirus B1 (コクサッキーウイルス) |
| 分子量 | 理論値: 29.122744 KDa |
| 配列 | 文字列: SPSAEECGYS DRVRSITLGN STITTQECAN VVVGYGVWPE YLKDNEATAE DQPTQPDVAT CRFYTLESVQ WMKNSAGWWW KLPDALSQM GLFGQNMQYH YLGRTGYTIH VQCNASKFHQ GCLLVVCVPE AEMGCSNLNN TPEFSELSGG DSARMFTDTQ V GESNAKKV ...文字列: SPSAEECGYS DRVRSITLGN STITTQECAN VVVGYGVWPE YLKDNEATAE DQPTQPDVAT CRFYTLESVQ WMKNSAGWWW KLPDALSQM GLFGQNMQYH YLGRTGYTIH VQCNASKFHQ GCLLVVCVPE AEMGCSNLNN TPEFSELSGG DSARMFTDTQ V GESNAKKV QTAVWNAGMG VGVGNLTIFP HQWINLRTNN SATLVMPYIN SVPMDNMFRH NNLTLMIIPF VPLNYSEGSS PY VPITVTI APMCAEYNGL RLASNQ UniProtKB: Genome polyprotein |
-分子 #5: VP3
| 分子 | 名称: VP3 / タイプ: protein_or_peptide / ID: 5 / コピー数: 1 / 光学異性体: LEVO / EC番号: picornain 2A |
|---|---|
| 由来(天然) | 生物種:  Coxsackievirus B1 (コクサッキーウイルス) Coxsackievirus B1 (コクサッキーウイルス) |
| 分子量 | 理論値: 26.328764 KDa |
| 配列 | 文字列: GLPVMTTPGS TQFLTSDDFQ SPSAMPQFDV TPEMQIPGRV NNLMEIAEVD SVVPVNNTED NVSSLKAYQI PVQSNSDNGK QVFGFPLQP GANNVLNRTL LGEILNYYTH WSGSIKLTFM FCGSAMATGK FLLAYSPPGA GVPKNRKDAM LGTHVIWDVG L QSSCVLCV ...文字列: GLPVMTTPGS TQFLTSDDFQ SPSAMPQFDV TPEMQIPGRV NNLMEIAEVD SVVPVNNTED NVSSLKAYQI PVQSNSDNGK QVFGFPLQP GANNVLNRTL LGEILNYYTH WSGSIKLTFM FCGSAMATGK FLLAYSPPGA GVPKNRKDAM LGTHVIWDVG L QSSCVLCV PWISQTHYRY VVEDEYTAAG YVTCWYQTNI VVPADVQSSC DILCFVSACN DFSVRMLKDT PFIRQDTFYQ UniProtKB: Genome polyprotein |
-分子 #6: Capsid protein VP4
| 分子 | 名称: Capsid protein VP4 / タイプ: protein_or_peptide / ID: 6 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Coxsackievirus B1 (コクサッキーウイルス) Coxsackievirus B1 (コクサッキーウイルス) |
| 分子量 | 理論値: 7.484246 KDa |
| 配列 | 文字列: MGAQVSTQKT GAHETGLNAS GNSVIHYTNI NYYKDAASNS ANRQDFTQDP GKFTEPVKDI MVKTMPALN UniProtKB: Genome polyprotein |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TECNAI F30 |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON III (4k x 4k) 検出モード: INTEGRATING / 平均電子線量: 60.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 3.5 µm / 最小 デフォーカス(公称値): 1.2 µm |
| 実験機器 |  モデル: Tecnai F30 / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー


























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)




















