[English] 日本語
 Yorodumi
Yorodumi- EMDB-32864: Structure of an assembly-line polyketide synthase module with typ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of an assembly-line polyketide synthase module with typical docking domain position | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | megaenzyme / HYDROLASE | |||||||||
| Biological species |  Streptomyces chartreusis NRRL 3882cha (bacteria) Streptomyces chartreusis NRRL 3882cha (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.55 Å | |||||||||
 Authors Authors | Wang J / Wang Z | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: C-N bond formation by a polyketide synthase. Authors: Jialiang Wang / Xiaojie Wang / Xixi Li / LiangLiang Kong / Zeqian Du / Dandan Li / Lixia Gou / Hao Wu / Wei Cao / Xiaozheng Wang / Shuangjun Lin / Ting Shi / Zixin Deng / Zhijun Wang / Jingdan Liang /  Abstract: Assembly-line polyketide synthases (PKSs) are molecular factories that produce diverse metabolites with wide-ranging biological activities. PKSs usually work by constructing and modifying the ...Assembly-line polyketide synthases (PKSs) are molecular factories that produce diverse metabolites with wide-ranging biological activities. PKSs usually work by constructing and modifying the polyketide backbone successively. Here, we present the cryo-EM structure of CalA3, a chain release PKS module without an ACP domain, and its structures with amidation or hydrolysis products. The domain organization reveals a unique "∞"-shaped dimeric architecture with five connected domains. The catalytic region tightly contacts the structural region, resulting in two stabilized chambers with nearly perfect symmetry while the N-terminal docking domain is flexible. The structures of the ketosynthase (KS) domain illustrate how the conserved key residues that canonically catalyze C-C bond formation can be tweaked to mediate C-N bond formation, revealing the engineering adaptability of assembly-line polyketide synthases for the production of novel pharmaceutical agents. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32864.map.gz emd_32864.map.gz | 49.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32864-v30.xml emd-32864-v30.xml emd-32864.xml emd-32864.xml | 14.4 KB 14.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_32864_fsc.xml emd_32864_fsc.xml | 8.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_32864.png emd_32864.png | 78.3 KB | ||
| Filedesc metadata |  emd-32864.cif.gz emd-32864.cif.gz | 3.9 KB | ||
| Others |  emd_32864_additional_1.map.gz emd_32864_additional_1.map.gz emd_32864_half_map_1.map.gz emd_32864_half_map_1.map.gz emd_32864_half_map_2.map.gz emd_32864_half_map_2.map.gz | 40.7 MB 40.8 MB 40.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32864 http://ftp.pdbj.org/pub/emdb/structures/EMD-32864 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32864 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32864 | HTTPS FTP |
-Validation report
| Summary document |  emd_32864_validation.pdf.gz emd_32864_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_32864_full_validation.pdf.gz emd_32864_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_32864_validation.xml.gz emd_32864_validation.xml.gz | 13.9 KB | Display | |
| Data in CIF |  emd_32864_validation.cif.gz emd_32864_validation.cif.gz | 19.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32864 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32864 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32864 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32864 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_32864.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32864.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
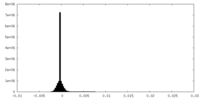
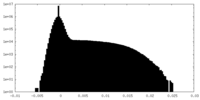
-Additional map: #1
| File | emd_32864_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
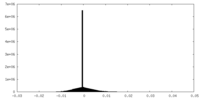
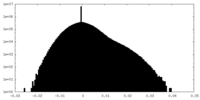
| Density Histograms |
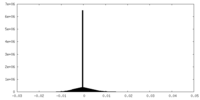
-Half map: #1
| File | emd_32864_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
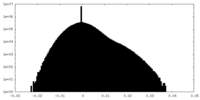
-Half map: #2
| File | emd_32864_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : an assembly-line polyketide synthase module containing KS-AT-DH-K...
| Entire | Name: an assembly-line polyketide synthase module containing KS-AT-DH-KR domains |
|---|---|
| Components |
|
-Supramolecule #1: an assembly-line polyketide synthase module containing KS-AT-DH-K...
| Supramolecule | Name: an assembly-line polyketide synthase module containing KS-AT-DH-KR domains type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Streptomyces chartreusis NRRL 3882cha (bacteria) Streptomyces chartreusis NRRL 3882cha (bacteria) |
| Molecular weight | Theoretical: 0.36 kDa/nm |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.9 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 48.6 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z
Z Y
Y X
X