+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of CD69-bound S1PR1 coupled to heterotrimeric Gi | ||||||||||||
 Map data Map data | Structure of CD69-bound S1PR1 coupled to heterotrimeric Gi | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | CD69 / S1PR1 / Sphingosine-1-phosphate / lymphocyte egress / GPCR / Guanine nucleotide-binding protein | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of T cell migration / cardiac muscle tissue growth involved in heart morphogenesis / blood vessel maturation / sphingolipid binding / sphingosine-1-phosphate receptor activity / Lysosphingolipid and LPA receptors / negative regulation of T-helper 17 cell lineage commitment / endothelial cell differentiation / heart trabecula morphogenesis / regulation of bone mineralization ...negative regulation of T cell migration / cardiac muscle tissue growth involved in heart morphogenesis / blood vessel maturation / sphingolipid binding / sphingosine-1-phosphate receptor activity / Lysosphingolipid and LPA receptors / negative regulation of T-helper 17 cell lineage commitment / endothelial cell differentiation / heart trabecula morphogenesis / regulation of bone mineralization / sphingosine-1-phosphate receptor signaling pathway / leukocyte chemotaxis / positive regulation of positive chemotaxis / regulation of bone resorption / negative regulation of stress fiber assembly / lamellipodium assembly / regulation of metabolic process / transmission of nerve impulse / molecular sequestering activity / regulation of cell adhesion / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration / Adenylate cyclase inhibitory pathway / D2 dopamine receptor binding / response to prostaglandin E / adenylate cyclase regulator activity / G protein-coupled serotonin receptor binding / adenylate cyclase-inhibiting serotonin receptor signaling pathway / positive regulation of smooth muscle cell proliferation / cellular response to forskolin / regulation of mitotic spindle organization / Regulation of insulin secretion / positive regulation of cholesterol biosynthetic process / negative regulation of insulin secretion / G protein-coupled receptor binding / G protein-coupled receptor activity / response to peptide hormone / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / brain development / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein beta/gamma-subunit complex binding / centriolar satellite / cellular response to xenobiotic stimulus / Olfactory Signaling Pathway / Activation of the phototransduction cascade / adenylate cyclase-activating G protein-coupled receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G protein-coupled acetylcholine receptor signaling pathway / neuron differentiation / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / chemotaxis / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through CDC42 / Glucagon signaling in metabolic regulation / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / ADP signalling through P2Y purinoceptor 12 / photoreceptor disc membrane / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / GDP binding / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / transmembrane signaling receptor activity / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / cell migration / GPER1 signaling / Inactivation, recovery and regulation of the phototransduction cascade / cellular response to prostaglandin E stimulus / G-protein beta-subunit binding / heterotrimeric G-protein complex / G alpha (12/13) signalling events / sensory perception of taste / presynapse / extracellular vesicle / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / retina development in camera-type eye / G protein activity / carbohydrate binding / GTPase binding / actin cytoskeleton organization / Ca2+ pathway / fibroblast proliferation / midbody / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / angiogenesis / cell cortex Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.15 Å | ||||||||||||
 Authors Authors | Chen H / Li X | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Elife / Year: 2023 Journal: Elife / Year: 2023Title: Transmembrane protein CD69 acts as an S1PR1 agonist. Authors: Hongwen Chen / Yu Qin / Marissa Chou / Jason G Cyster / Xiaochun Li /  Abstract: The activation of Sphingosine-1-phosphate receptor 1 (S1PR1) by S1P promotes lymphocyte egress from lymphoid organs, a process critical for immune surveillance and T cell effector activity. Multiple ...The activation of Sphingosine-1-phosphate receptor 1 (S1PR1) by S1P promotes lymphocyte egress from lymphoid organs, a process critical for immune surveillance and T cell effector activity. Multiple drugs that inhibit S1PR1 function are in use clinically for the treatment of autoimmune diseases. Cluster of Differentiation 69 (CD69) is an endogenous negative regulator of lymphocyte egress that interacts with S1PR1 in cis to facilitate internalization and degradation of the receptor. The mechanism by which CD69 causes S1PR1 internalization has been unclear. Moreover, although there are numerous class A GPCR structures determined with different small molecule agonists bound, it remains unknown whether a transmembrane protein per se can act as a class A GPCR agonist. Here, we present the cryo-EM structure of CD69-bound S1PR1 coupled to the heterotrimeric G complex. The transmembrane helix (TM) of one protomer of CD69 homodimer contacts the S1PR1-TM4. This interaction allosterically induces the movement of S1PR1-TMs 5-6, directly activating the receptor to engage the heterotrimeric G. Mutations in key residues at the interface affect the interactions between CD69 and S1PR1, as well as reduce the receptor internalization. Thus, our structural findings along with functional analyses demonstrate that CD69 acts in cis as a protein agonist of S1PR1, thereby promoting G-dependent S1PR1 internalization, loss of S1P gradient sensing, and inhibition of lymphocyte egress. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29861.map.gz emd_29861.map.gz | 84.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29861-v30.xml emd-29861-v30.xml emd-29861.xml emd-29861.xml | 26.6 KB 26.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_29861_fsc.xml emd_29861_fsc.xml | 9.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_29861.png emd_29861.png | 47.2 KB | ||
| Filedesc metadata |  emd-29861.cif.gz emd-29861.cif.gz | 7.9 KB | ||
| Others |  emd_29861_half_map_1.map.gz emd_29861_half_map_1.map.gz emd_29861_half_map_2.map.gz emd_29861_half_map_2.map.gz | 71.2 MB 71.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29861 http://ftp.pdbj.org/pub/emdb/structures/EMD-29861 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29861 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29861 | HTTPS FTP |
-Validation report
| Summary document |  emd_29861_validation.pdf.gz emd_29861_validation.pdf.gz | 915.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29861_full_validation.pdf.gz emd_29861_full_validation.pdf.gz | 915.3 KB | Display | |
| Data in XML |  emd_29861_validation.xml.gz emd_29861_validation.xml.gz | 17.6 KB | Display | |
| Data in CIF |  emd_29861_validation.cif.gz emd_29861_validation.cif.gz | 22.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29861 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29861 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29861 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29861 | HTTPS FTP |
-Related structure data
| Related structure data |  8g94MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29861.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29861.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of CD69-bound S1PR1 coupled to heterotrimeric Gi | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||
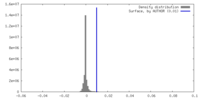
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half Map 1
| File | emd_29861_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
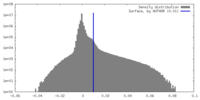
| Density Histograms |
-Half map: Half Map 2
| File | emd_29861_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of CD69-bound S1PR1 coupled to heterotrimeric Gi
| Entire | Name: Complex of CD69-bound S1PR1 coupled to heterotrimeric Gi |
|---|---|
| Components |
|
-Supramolecule #1: Complex of CD69-bound S1PR1 coupled to heterotrimeric Gi
| Supramolecule | Name: Complex of CD69-bound S1PR1 coupled to heterotrimeric Gi type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 200 KDa |
-Macromolecule #1: Sphingosine 1-phosphate receptor 1
| Macromolecule | Name: Sphingosine 1-phosphate receptor 1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.083781 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGPTSVPLVK AHRSSVSDYV NYDIIVRHYN YTGKLNISAD KENSIKLTSV VFILICCFII LENIFVLLTI WKTKKFHRPM YYFIGNLAL SDLLAGVAYT ANLLLSGATT YKLTPAQWFL REGSMFVALS ASVFSLLAIA IERYITMLKM KLHNGSNNFR L FLLISACW ...String: MGPTSVPLVK AHRSSVSDYV NYDIIVRHYN YTGKLNISAD KENSIKLTSV VFILICCFII LENIFVLLTI WKTKKFHRPM YYFIGNLAL SDLLAGVAYT ANLLLSGATT YKLTPAQWFL REGSMFVALS ASVFSLLAIA IERYITMLKM KLHNGSNNFR L FLLISACW VISLILGGLP IMGWNCISAL SSCSTVLPLY HKHYILFCTT VFTLLLLSIV ILYCRIYSLV RTRSRRLTFR KN ISKASRS SEKSLALLKT VIIVLSVFIA CWAPLFILLL LDVGCKVKTC DILFRAEYFL VLAVLNSGTN PIIYTLTNKE MRR AFIRIM SCCKCPSGDS AGKFKRPIIA GDYKDDDDK UniProtKB: Sphingosine 1-phosphate receptor 1 |
-Macromolecule #2: Guanine nucleotide-binding protein G(i) subunit alpha-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(i) subunit alpha-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.445059 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTTGIVETH FTFKDLHFKM FDVGAQRSER KKWIHCFEGV TAIIFCVALS DYDLVLAEDE EM NRMHESM KLFDSICNNK WFTDTSIILF LNKKDLFEEK IKKSPLTICY PEYAGSNTYE EAAAYIQCQF EDLNKRKDTK EIY THFTCS TDTKNVQFVF DAVTDVIIKN NLKDCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.728152 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GPGSSGSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD ...String: GPGSSGSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD TTCALWDIET GQQTTTFTGH TGDVMSLSLA PDTRLFVSGA CDASAKLWDV REGMCRQTFT GHESDINAIC FF PNGNAFA TGSDDATCRL FDLRADQELM TYSHDNIICG ITSVSFSKSG RLLLAGYDDF NCNVWDALKA DRAGVLAGHD NRV SCLGVT DDGMAVATGS WDSFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: scFv16
| Macromolecule | Name: scFv16 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 27.784896 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS ...String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS NGNTYLYWFL QRPGQSPQLL IYRMSNLASG VPDRFSGSGS GTAFTLTISR LEAEDVGVYY CMQHLEYPLT FG AGTKLEL KAAAHHHHHH HH |
-Macromolecule #6: Early activation antigen CD69
| Macromolecule | Name: Early activation antigen CD69 / type: protein_or_peptide / ID: 6 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.912793 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MASENCFVAE NSSLHPESGQ ENDATSPHFS TRHEGSFQVP VLCAVMNVVF ITILIIALIA LSVGQYNCPG QYTFSMPSDS HVSSCSEDW VGYQRKCYFI STVKRSWTSA QNACSEHGAT LAVIDSEKDM NFLKRYAGRE EHWVGLKKEP GHPWKWSNGK E FNNWFNVT ...String: MASENCFVAE NSSLHPESGQ ENDATSPHFS TRHEGSFQVP VLCAVMNVVF ITILIIALIA LSVGQYNCPG QYTFSMPSDS HVSSCSEDW VGYQRKCYFI STVKRSWTSA QNACSEHGAT LAVIDSEKDM NFLKRYAGRE EHWVGLKKEP GHPWKWSNGK E FNNWFNVT GSDKCVFLKN TEVSSMECEK NLYWICNKPY KGSASWSHPQ FEK UniProtKB: Early activation antigen CD69 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 10 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)