[English] 日本語
 Yorodumi
Yorodumi- EMDB-27344: cryo-EM structure of TRPM3 ion channel in complex with Gbg, tethe... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | cryo-EM structure of TRPM3 ion channel in complex with Gbg, tethered by ALFA-nanobody | |||||||||
 Map data Map data | sharpened map with a B-factor of 112.4 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TRPM3 / ion channel / Gbg / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationzinc ion transmembrane transporter activity / zinc ion transmembrane transport / temperature-gated ion channel activity / sodium ion transport / monoatomic cation transport / phosphatidylinositol-4,5-bisphosphate binding / calcium ion transmembrane transport / calcium channel activity / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway ...zinc ion transmembrane transporter activity / zinc ion transmembrane transport / temperature-gated ion channel activity / sodium ion transport / monoatomic cation transport / phosphatidylinositol-4,5-bisphosphate binding / calcium ion transmembrane transport / calcium channel activity / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G protein-coupled acetylcholine receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / G beta:gamma signalling through BTK / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / G-protein beta-subunit binding / extracellular vesicle / sensory perception of taste / Thrombin signalling through proteinase activated receptors (PARs) / signaling receptor complex adaptor activity / retina development in camera-type eye / GTPase binding / fibroblast proliferation / Ca2+ pathway / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / G alpha (i) signalling events / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / protein homotetramerization / G alpha (q) signalling events / Ras protein signal transduction / calmodulin binding / Extra-nuclear estrogen signaling / cell population proliferation / G protein-coupled receptor signaling pathway / lysosomal membrane / GTPase activity / synapse / protein-containing complex binding / signal transduction / extracellular exosome / membrane / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.7 Å | |||||||||
 Authors Authors | Zhao C / MacKinnon R | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Neuron / Year: 2023 Journal: Neuron / Year: 2023Title: Structural and functional analyses of a GPCR-inhibited ion channel TRPM3. Authors: Chen Zhao / Roderick MacKinnon /  Abstract: G-protein coupled receptors (GPCRs) govern the physiological response to stimuli by modulating the activity of downstream effectors, including ion channels. TRPM3 is an ion channel inhibited by GPCRs ...G-protein coupled receptors (GPCRs) govern the physiological response to stimuli by modulating the activity of downstream effectors, including ion channels. TRPM3 is an ion channel inhibited by GPCRs through direct interaction with G protein (Gβγ) released upon their activation. This GPCR-TRPM3 signaling pathway contributes to the analgesic effect of morphine. Here, we characterized Gβγ inhibition of TRPM3 using electrophysiology and single particle cryo-electron microscopy (cryo-EM). From electrophysiology, we obtained a half inhibition constant (IC50) of ∼240 nM. Using cryo-EM, we determined structures of mouse TRPM3 expressed in human cells with and without Gβγ and with and without PIP, a lipid required for TRPM3 activity, at resolutions of 2.7-4.7 Å. Gβγ-TRPM3 interfaces vary depending on PIP occupancy; however, in all cases, Gβγ appears loosely attached to TRPM3. The IC50 in electrophysiology experiments raises the possibility that additional unknown factors may stabilize the TRPM3-Gβγ complex. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27344.map.gz emd_27344.map.gz | 16.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27344-v30.xml emd-27344-v30.xml emd-27344.xml emd-27344.xml | 21.6 KB 21.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27344_fsc.xml emd_27344_fsc.xml | 15.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_27344.png emd_27344.png | 101.6 KB | ||
| Filedesc metadata |  emd-27344.cif.gz emd-27344.cif.gz | 7.4 KB | ||
| Others |  emd_27344_half_map_1.map.gz emd_27344_half_map_1.map.gz emd_27344_half_map_2.map.gz emd_27344_half_map_2.map.gz | 275.5 MB 275.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27344 http://ftp.pdbj.org/pub/emdb/structures/EMD-27344 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27344 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27344 | HTTPS FTP |
-Related structure data
| Related structure data |  8ddwMC  8ddqC  8ddrC  8ddsC  8ddtC  8dduC  8ddvC  8ddxC  8ed7C  8ed8C  8ed9C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27344.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27344.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map with a B-factor of 112.4 | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
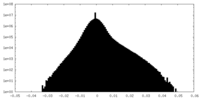
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: half map 1
| File | emd_27344_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
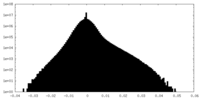
| Density Histograms |
-Half map: half map 2
| File | emd_27344_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
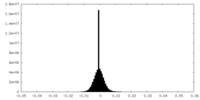
| Density Histograms |
- Sample components
Sample components
-Entire : TRPM3
| Entire | Name: TRPM3 |
|---|---|
| Components |
|
-Supramolecule #1: TRPM3
| Supramolecule | Name: TRPM3 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Transient receptor potential cation channel, subfamily M, member 3
| Macromolecule | Name: Transient receptor potential cation channel, subfamily M, member 3 type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 157.812922 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGKKWRDAGE LERGCSDRED SAESRRRSRS ASRGRFAESW KRLSSKQGST KRSGLPAQQT PAQKSWIERA FYKRECVHII PSTKDPHRC CCGRLIGQHV GLTPSISVLQ NEKNESRLSR NDIQSEKWSI SKHTQLSPTD AFGTIEFQGG GHSNKAMYVR V SFDTKPDL ...String: MGKKWRDAGE LERGCSDRED SAESRRRSRS ASRGRFAESW KRLSSKQGST KRSGLPAQQT PAQKSWIERA FYKRECVHII PSTKDPHRC CCGRLIGQHV GLTPSISVLQ NEKNESRLSR NDIQSEKWSI SKHTQLSPTD AFGTIEFQGG GHSNKAMYVR V SFDTKPDL LLHLMTKEWQ LELPKLLISV HGGLQNFELQ PKLKQVFGKG LIKAAMTTGA WIFTGGVNTG VIRHVGDALK DH ASKSRGK ICTIGIAPWG IVENQEDLIG RDVVRPYQTM SNPMSKLTVL NSMHSHFILA DNGTTGKYGA EVKLRRQLEK HIS LQKINT RIGQGVPVVA LIVEGGPNVI SIVLEYLRDT PPVPVVVCDG SGRASDILAF GHKYSEEGGL INESLRDQLL VTIQ KTFTY TRTQAQHLFI ILMECMKKKE LITVFRMGSE GHQDIDLAIL TALLKGANAS APDQLSLALA WNRVDIARSQ IFIYG QQWP VGSLEQAMLD ALVLDRVDFV KLLIENGVSM HRFLTISRLE ELYNTRHGPS NTLYHLVRDV KKGNLPPDYR ISLIDI GLV IEYLMGGAYR CNYTRKRFRT LYHNLFGPKR PKALKLLGME DDIPLRRGRK TTKKREEEVD IDLDDPEINH FPFPFHE LM VWAVLMKRQK MALFFWQHGE EAMAKALVAC KLCKAMAHEA SENDMVDDIS QELNHNSRDF GQLAVELLDQ SYKQDEQL A MKLLTYELKN WSNATCLQLA VAAKHRDFIA HTCSQMLLTD MWMGRLRMRK NSGLKVILGI LLPPSILSLE FKNKDDMPY MTQAQEIHLQ EKEPEEPEKP TKEKDEEDME LTAMLGRSNG ESSRKKDEEE VQSRHRLIPV GRKIYEFYNA PIVKFWFYTL AYIGYLMLF NYIVLVKMER WPSTQEWIVI SYIFTLGIEK MREILMSEPG KLLQKVKVWL QEYWNVTDLI AILLFSVGMI L RLQDQPFR SDGRVIYCVN IIYWYIRLLD IFGVNKYLGP YVMMIGKMMI DMMYFVIIML VVLMSFGVAR QAILFPNEEP SW KLAKNIF YMPYWMIYGE VFADQIDPPC GQNETREDGK TIQLPPCKTG AWIVPAIMAC YLLVANILLV NLLIAVFNNT FFE VKSISN QVWKFQRYQL IMTFHERPVL PPPLIIFSHM TMIFQHVCCR WRKHESDQDE RDYGLKLFIT DDELKKVHDF EEQC IEEYF REKDDRFNSS NDERIRVTSE RVENMSMRLE EVNEREHSMK ASLQTVDIRL AQLEDLIGRM ATALERLTGL ERAES NKIR SRTSSDCTDA AYIVRQSSFN SQEGNTFKLQ ESIDPAGEET ISPTSPTLMP RMRSHSFYSV NVKDKGGIEK LESIFK ERS LSLHRATS UniProtKB: Transient receptor potential cation channel subfamily M member 3 |
-Macromolecule #2: Unidentified segment at the N-terminus of TRPM3
| Macromolecule | Name: Unidentified segment at the N-terminus of TRPM3 / type: protein_or_peptide / ID: 2 Details: a segment at the N-terminus of TRPM3 whose sequence cannot be identified from the cryo-EM density Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.464797 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.285734 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: SELDQLRQEA EQLKNQIRDA RKACADATLS QITNNIDPVG RIQMRTRRTL RGHLAKIYAM HWGTDSRLLV SASQDGKLII WDSYTTNKV HAIPLRSSWV MTCAYAPSGN YVACGGLDNI CSIYNLKTRE GNVRVSRELA GHTGYLSCCR FLDDNQIVTS S GDTTCALW ...String: SELDQLRQEA EQLKNQIRDA RKACADATLS QITNNIDPVG RIQMRTRRTL RGHLAKIYAM HWGTDSRLLV SASQDGKLII WDSYTTNKV HAIPLRSSWV MTCAYAPSGN YVACGGLDNI CSIYNLKTRE GNVRVSRELA GHTGYLSCCR FLDDNQIVTS S GDTTCALW DIETGQQTTT FTGHTGDVMS LSLAPDTRLF VSGACDASAK LWDVREGMCR QTFTGHESDI NAICFFPNGN AF ATGSDDA TCRLFDLRAD QELMTYSHDN IICGITSVSF SKSGRLLLAG YDDFNCNVWD ALKADRAGVL AGHDNRVSCL GVT DDGMAV ATGSWDSFLK IWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.713882 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: ASNNTASIAQ ARKLVEQLKM EANIDRIKVS KAAADLMAYC EAHAKEDPLL TPVPASENPF REKKFFSAIL UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 51.4 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)