[English] 日本語
 Yorodumi
Yorodumi- EMDB-27288: Human PRPS1 with Phosphate, ATP, and R5P; Hexamer with resolved c... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human PRPS1 with Phosphate, ATP, and R5P; Hexamer with resolved catalytic loops | |||||||||
 Map data Map data | Human PRPS1 with Phosphate, ATP, and R5P; Hexamer Centered with resolved catalytic loops | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | phosphoribosyl pyrophosphate synthetase / ATP and R5P substrates / catalytic loop / TRANSFERASE | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.1 Å | |||||||||
 Authors Authors | Hvorecny KL / Kollman JM | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Human PRPS1 filaments stabilize allosteric sites to regulate activity. Authors: Kelli L Hvorecny / Kenzee Hargett / Joel D Quispe / Justin M Kollman /  Abstract: The universally conserved enzyme phosphoribosyl pyrophosphate synthetase (PRPS) assembles filaments in evolutionarily diverse organisms. PRPS is a key regulator of nucleotide metabolism, and ...The universally conserved enzyme phosphoribosyl pyrophosphate synthetase (PRPS) assembles filaments in evolutionarily diverse organisms. PRPS is a key regulator of nucleotide metabolism, and mutations in the human enzyme PRPS1 lead to a spectrum of diseases. Here we determine structures of human PRPS1 filaments in active and inhibited states, with fixed assembly contacts accommodating both conformations. The conserved assembly interface stabilizes the binding site for the essential activator phosphate, increasing activity in the filament. Some disease mutations alter assembly, supporting the link between filament stability and activity. Structures of active PRPS1 filaments turning over substrate also reveal coupling of catalysis in one active site with product release in an adjacent site. PRPS1 filaments therefore provide an additional layer of allosteric control, conserved throughout evolution, with likely impact on metabolic homeostasis. Stabilization of allosteric binding sites by polymerization adds to the growing diversity of assembly-based enzyme regulatory mechanisms. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27288.map.gz emd_27288.map.gz | 13.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27288-v30.xml emd-27288-v30.xml emd-27288.xml emd-27288.xml | 15.2 KB 15.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_27288.png emd_27288.png | 118.5 KB | ||
| Filedesc metadata |  emd-27288.cif.gz emd-27288.cif.gz | 5 KB | ||
| Others |  emd_27288_half_map_1.map.gz emd_27288_half_map_1.map.gz emd_27288_half_map_2.map.gz emd_27288_half_map_2.map.gz | 97.8 MB 97.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27288 http://ftp.pdbj.org/pub/emdb/structures/EMD-27288 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27288 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27288 | HTTPS FTP |
-Validation report
| Summary document |  emd_27288_validation.pdf.gz emd_27288_validation.pdf.gz | 802.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_27288_full_validation.pdf.gz emd_27288_full_validation.pdf.gz | 802.2 KB | Display | |
| Data in XML |  emd_27288_validation.xml.gz emd_27288_validation.xml.gz | 13.9 KB | Display | |
| Data in CIF |  emd_27288_validation.cif.gz emd_27288_validation.cif.gz | 16.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27288 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27288 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27288 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27288 | HTTPS FTP |
-Related structure data
| Related structure data |  8dbcC  8dbdC  8dbeC  8dbfC  8dbgC  8dbhC  8dbiC  8dbjC  8dbkC  8dblC  8dbmC  8dbnC  8dboC C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_27288.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27288.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human PRPS1 with Phosphate, ATP, and R5P; Hexamer Centered with resolved catalytic loops | ||||||||||||||||||||||||||||||||||||
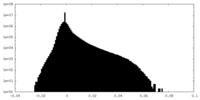
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.827 Å | ||||||||||||||||||||||||||||||||||||
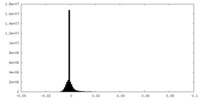
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Human PRPS1 with Phosphate, ATP, and R5P; Hexamer...
| File | emd_27288_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human PRPS1 with Phosphate, ATP, and R5P; Hexamer Centered with resolved catalytic loops half 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
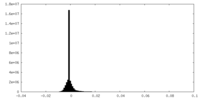
| Density Histograms |
-Half map: Human PRPS1 with Phosphate, ATP, and R5P; Hexamer...
| File | emd_27288_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human PRPS1 with Phosphate, ATP, and R5P; Hexamer Centered with resolved catalytic loops half 1 | ||||||||||||
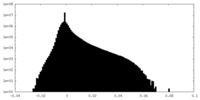
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Hexamer of phosphoribosyl pyrophosphate synthetase 1 with phospha...
| Entire | Name: Hexamer of phosphoribosyl pyrophosphate synthetase 1 with phosphate, ATP, and R5P; resolved catalytic loops |
|---|---|
| Components |
|
-Supramolecule #1: Hexamer of phosphoribosyl pyrophosphate synthetase 1 with phospha...
| Supramolecule | Name: Hexamer of phosphoribosyl pyrophosphate synthetase 1 with phosphate, ATP, and R5P; resolved catalytic loops type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Six copies of PRPS1 assemble to form a hexamer; hexamers assembly linearly to form filaments. Here, two of the catalytic loops are well resolved. |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Human Phosphoribosyl Pyrophosphate Synthetase 1
| Macromolecule | Name: Human Phosphoribosyl Pyrophosphate Synthetase 1 / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
| Sequence | String: MPNIKIFSGS SHQDLSQKIA DRLGLELGKV VTKKFSNQET CVEIGESVRG EDVYIVQSGC GEINDNLME LLIMINACKI ASASRVTAVI PCFPYARQDK KDKSRAPISA KLVANMLSVA G ADHIITMD LHASQIQGFF DIPVDNLYAE PAVLKWIREN ISEWRNCTIV ...String: MPNIKIFSGS SHQDLSQKIA DRLGLELGKV VTKKFSNQET CVEIGESVRG EDVYIVQSGC GEINDNLME LLIMINACKI ASASRVTAVI PCFPYARQDK KDKSRAPISA KLVANMLSVA G ADHIITMD LHASQIQGFF DIPVDNLYAE PAVLKWIREN ISEWRNCTIV SPDAGGAKRV TS IADRLNV DFALIHKERK KANEVDRMVL VGDVKDRVAI LVDDMADTCG TICHAADKLL SAG ATRVYA ILTHGIFSGP AISRINNACF EAVVVTNTIP QEDKMKHCSK IQVIDISMIL AEAI RRTHN GESVSYLFSH VPL |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 |
|---|---|
| Grid | Model: C-flat / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Instrument: HOMEMADE PLUNGER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 90.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.75 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 2.1 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: PHENIX (ver. 1.18) / Number images used: 912162 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC (ver. 3) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.1) |
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)