+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | gRAMP-TPR-CHAT match PFS target RNA(Craspase) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CRISPR / GRAMP / RNA BINDING PROTEIN / Craspase / RNA BINDING PROTEIN-RNA complex | |||||||||
| Function / homology | CHAT domain / CHAT domain / : / CRISPR type III-associated protein / RAMP superfamily / defense response to virus / RAMP superfamily protein / CHAT domain protein Function and homology information Function and homology information | |||||||||
| Biological species |  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Hu C / Nam KH / Schuler G / Ke A | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: Craspase is a CRISPR RNA-guided, RNA-activated protease. Authors: Chunyi Hu / Sam P B van Beljouw / Ki Hyun Nam / Gabriel Schuler / Fran Ding / Yanru Cui / Alicia Rodríguez-Molina / Anna C Haagsma / Menno Valk / Martin Pabst / Stan J J Brouns / Ailong Ke /    Abstract: The CRISPR-Cas type III-E RNA-targeting effector complex gRAMP/Cas7-11 is associated with a caspase-like protein (TPR-CHAT/Csx29) to form Craspase (CRISPR-guided caspase). Here, we use cryo-electron ...The CRISPR-Cas type III-E RNA-targeting effector complex gRAMP/Cas7-11 is associated with a caspase-like protein (TPR-CHAT/Csx29) to form Craspase (CRISPR-guided caspase). Here, we use cryo-electron microscopy snapshots of Craspase to explain its target RNA cleavage and protease activation mechanisms. Target-guide pairing extending into the 5' region of the guide RNA displaces a gating loop in gRAMP, which triggers an extensive conformational relay that allosterically aligns the protease catalytic dyad and opens an amino acid side-chain-binding pocket. We further define Csx30 as the endogenous protein substrate that is site-specifically proteolyzed by RNA-activated Craspase. This protease activity is switched off by target RNA cleavage by gRAMP and is not activated by RNA targets containing a matching protospacer flanking sequence. We thus conclude that Craspase is a target RNA-activated protease with self-regulatory capacity. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27262.map.gz emd_27262.map.gz | 48.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27262-v30.xml emd-27262-v30.xml emd-27262.xml emd-27262.xml | 23.5 KB 23.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_27262.png emd_27262.png | 118.3 KB | ||
| Filedesc metadata |  emd-27262.cif.gz emd-27262.cif.gz | 7.5 KB | ||
| Others |  emd_27262_additional_1.map.gz emd_27262_additional_1.map.gz emd_27262_half_map_1.map.gz emd_27262_half_map_1.map.gz emd_27262_half_map_2.map.gz emd_27262_half_map_2.map.gz | 37.1 MB 77.7 MB 77.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27262 http://ftp.pdbj.org/pub/emdb/structures/EMD-27262 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27262 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27262 | HTTPS FTP |
-Related structure data
| Related structure data |  8d9hMC  8d8nC  8d97C  8d9eC  8d9fC  8d9gC  8d9iC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_27262.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27262.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_27262_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
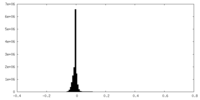
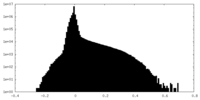
| Density Histograms |
-Half map: #1
| File | emd_27262_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
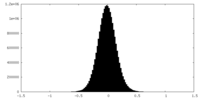
| Density Histograms |
-Half map: #2
| File | emd_27262_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
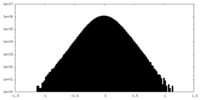
| Density Histograms |
- Sample components
Sample components
-Entire : gRAMP-TPR-CHAT with match PFS target RNA Craspase
| Entire | Name: gRAMP-TPR-CHAT with match PFS target RNA Craspase |
|---|---|
| Components |
|
-Supramolecule #1: gRAMP-TPR-CHAT with match PFS target RNA Craspase
| Supramolecule | Name: gRAMP-TPR-CHAT with match PFS target RNA Craspase / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) |
-Macromolecule #1: RAMP superfamily protein
| Macromolecule | Name: RAMP superfamily protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) |
| Molecular weight | Theoretical: 145.301531 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MNITVELTFF EPYRLVEWFD WDARKKSHSA MRGQAFAQWT WKGKGRTAGK SFITGTLVRS AVIKAVEELL SLNNGKWEGV PCCNGSFQT DESKGKKPSF LRKRHTLQWQ ANNKNICDKE EACPFCILLG RFDNAGKVHE RNKDYDIHFS NFDLDHKQEK N DLRLVDIA ...String: MNITVELTFF EPYRLVEWFD WDARKKSHSA MRGQAFAQWT WKGKGRTAGK SFITGTLVRS AVIKAVEELL SLNNGKWEGV PCCNGSFQT DESKGKKPSF LRKRHTLQWQ ANNKNICDKE EACPFCILLG RFDNAGKVHE RNKDYDIHFS NFDLDHKQEK N DLRLVDIA SGRILNRVDF DTGKAKDYFR TWEADYETYG TYTGRITLRN EHAKKLLLAS LGFVDKLCGA LCRIEVIKDH ND ELRKQAE VIVEAFKQND KLEKIRILAD AIRTLRLHGE GVIEKDELPD GKEERDKGHH LWDIKVQGTA LRTKLKELWQ SNK DIGWRK FTEMLGSNLY LIYKKETGGV STEYYSKAHD SEGSDLFIPV TPPEGIETKE WIIVGRLKAA TPFYFGVQQP SDSI PGKEI NEHTSFNILL DKENRYRIPR SALRGALRRD LRTAFGSGCN VSLGGQILCN CKVCIEMRRI TLKDSVSDFS EPPEI RYRI AKNPGTATVE DGSLFDIEVG PEGLTFPFVL RYRGHKFPEQ LSSVIRYWEE NDGKNGMAWL GGLDSTGKGR FALKDI KIF EWDLNQKINE YIKERGMRGK EKELLEMGES SLPDGLIPYK FFEERECLFP YKENLKPQWS EVQYTIEVGS PLLTADT IS ALTEPGNRDA IAYKKRVYND GNNAIEPEPR FAVKSETHRG IFRTAVGRRT GDLGKEDHED CTCDMCIIFG NEHESSKI R FEDLELINGN EFEKLEKHID HVAIDRFTGG ALDKAKFDTY PLAGSPKKPL KLKGRFWIKK GFSGDHKLLI TTALSDIRD GLYPLGSKGG VGYGWVAGIS IDDNVPDDFK EMINKTNNDY VHPGHQSPKQ DHKNKNIYYP HYFLDSGSKV YREKDIITHE EFTEELLSG KINCKLETLT PLIIPDTSDE NGLKLQGNKP GHKNYKFFNI NGELMIPGSE LRGMLRTHFE ALTKSCFAIF G EDSTLSWA SKTLGGKLDK ALHPCTGLSD GLCPGCHLFG TTDYKGRVKF GFAKYENGPE WLITRGNNPE RSLTLGVLES PR PAFSIPD DESEIPGRKF YLHHNGWRII RQKQLEIRET VQPERNVTTE VMDKGNVFSF DVRFENLREW ELGLLLQSLD PGK NIAHKL GKGKPYGFGS VKIKIDSLHT FKINSNNDKI KRVPQSDIRE YINKGYQKLI EWSGNNSIQK GNVLPQWHVI PHID KLYKL LWVPFLNDSK LEPDVRYPVL NEESKGYIEG SDYTYKKLGD KDNLPYKTRV KGLTTPWSPW NPFQV UniProtKB: RAMP superfamily protein, RAMP superfamily protein |
-Macromolecule #4: CHAT domain protein
| Macromolecule | Name: CHAT domain protein / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) |
| Molecular weight | Theoretical: 76.802992 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: TREDIDRKEA ERLLDEAFNP RTKPVDRKKI INSALKILIG LYKEKKDDLT SASFISIARA YYLVSITILP KGTTIPEKKK EALRKGIEF IDRAINKFNG SILDSQRAFR IKSVLSIEFN RIDREKCDNI KLKNLLNEAV DKGCTDFDTY EWDIQIAIRL C ELGVDMEG ...String: TREDIDRKEA ERLLDEAFNP RTKPVDRKKI INSALKILIG LYKEKKDDLT SASFISIARA YYLVSITILP KGTTIPEKKK EALRKGIEF IDRAINKFNG SILDSQRAFR IKSVLSIEFN RIDREKCDNI KLKNLLNEAV DKGCTDFDTY EWDIQIAIRL C ELGVDMEG HFDNLIKSNK ANDLQKAKAY YFIKKDDHKA KEHMDKCTAS LKYTPCSHRL WDETVGFIER LKGDSSTLWR DF AIKTYRS CRVQEKETGT LRLRWYWSRH RVLYDMAFLA VKEQADVNVK QAKIKKLAEI SDSLKSRFSL RLSDMEKMPK SDD ESNHEF KKFLDKCVTA YQDGYVIKLL ELTQVPEGWV VVHFYLNKLE GMGNAIVFDK CANSWQYKEF QYKELFEVFL TWQA NYNLY KENAAEHLVT LCKKIGETMP FLFCDNFIPN GKDVLFVPHD FLHRLPLHGS IENKTNGKLF LENHSCCYLP AWSFA SEKE ASTSDEYVLL KNFDQGHFET LQNNQIWGTQ SVKDGASSDD LENIRNNPRL LTILCHGEAN MSNPFRSMLK LANGGI TYL EILNSVKGLK GSQVILGACE TDLVPPLSHY SVATALLLIG AAGVVGTMWK VRSNKTKSLI EWKLENIEYK LNEWQKE TG GAAYKDHPPT FYRSIAFRSI GFPL UniProtKB: CHAT domain protein |
-Macromolecule #2: RNA (36-MER)
| Macromolecule | Name: RNA (36-MER) / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) |
| Molecular weight | Theoretical: 11.423787 KDa |
| Sequence | String: GACUUAAUGU CACGGUACCC AAUUUUCUGC CCCGGA |
-Macromolecule #3: RNA (5'-R(P*UP*CP*CP*GP*GP*GP*GP*CP*AP*GP*AP*AP*AP*AP*UP*UP*GP*GP...
| Macromolecule | Name: RNA (5'-R(P*UP*CP*CP*GP*GP*GP*GP*CP*AP*GP*AP*AP*AP*AP*UP*UP*GP*GP*GP*U)-3') type: rna / ID: 3 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Candidatus Scalindua brodae (bacteria) Candidatus Scalindua brodae (bacteria) |
| Molecular weight | Theoretical: 6.502928 KDa |
| Sequence | String: UCCGGGGCAG AAAAUUGGGU |
-Macromolecule #5: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 5 / Number of copies: 4 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #6: PHOSPHATE ION
| Macromolecule | Name: PHOSPHATE ION / type: ligand / ID: 6 / Number of copies: 3 / Formula: PO4 |
|---|---|
| Molecular weight | Theoretical: 94.971 Da |
| Chemical component information |  ChemComp-PO4: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)