+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2694 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MAPPING THE DEUBIQUITINATION MODULE WITHIN THE SAGA COMPLEX | |||||||||
 Map data Map data | Structure of mutant SAGA complex (Sgf73 deletion) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | SAGA / Eukaryotic Transcription / Deubiquitination module | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 25.8 Å | |||||||||
 Authors Authors | Durand A / Bonnet J / Fournier M / Schultz P | |||||||||
 Citation Citation |  Journal: Structure / Year: 2014 Journal: Structure / Year: 2014Title: Mapping the deubiquitination module within the SAGA complex. Abstract: The molecular organization of the yeast transcriptional coactivator Spt-Ada-Gcn5 acetyltransferase (SAGA) was analyzed by single-particle electron microscopy. Complete or partial deletion of the ...The molecular organization of the yeast transcriptional coactivator Spt-Ada-Gcn5 acetyltransferase (SAGA) was analyzed by single-particle electron microscopy. Complete or partial deletion of the Sgf73 subunit disconnects the deubiquitination (DUB) module from SAGA and favors in our conditions the cleavage of the C-terminal ends of the Spt7 subunit and the loss of the Spt8 subunit. The structural comparison of the wild-type SAGA with two deletion mutants positioned the DUB module and enabled the fitting of the available atomic models. The localization of the DUB module close to Gcn5 defines a chromatin-binding interface within SAGA, which could be demonstrated by the binding of nucleosome core particles. The TATA-box binding protein (TBP)-interacting subunit Spt8 was found to be located close to the DUB but in a different domain than Spt3, also known to contact TBP. A flexible protein arm brings both subunits close enough to interact simultaneously with TBP. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2694.map.gz emd_2694.map.gz | 2.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2694-v30.xml emd-2694-v30.xml emd-2694.xml emd-2694.xml | 8.2 KB 8.2 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD-2694-12638.png EMD-2694-12638.png | 96.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2694 http://ftp.pdbj.org/pub/emdb/structures/EMD-2694 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2694 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2694 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2694.map.gz / Format: CCP4 / Size: 51.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2694.map.gz / Format: CCP4 / Size: 51.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of mutant SAGA complex (Sgf73 deletion) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.05 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
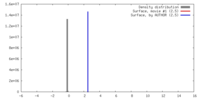
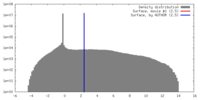
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : S. cerevisiae SAGA complex (Sgf73 deletion)
| Entire | Name: S. cerevisiae SAGA complex (Sgf73 deletion) |
|---|---|
| Components |
|
-Supramolecule #1000: S. cerevisiae SAGA complex (Sgf73 deletion)
| Supramolecule | Name: S. cerevisiae SAGA complex (Sgf73 deletion) / type: sample / ID: 1000 / Details: The sample was monodisperse / Number unique components: 1 |
|---|---|
| Molecular weight | Theoretical: 1.8 MDa |
-Macromolecule #1: SAGA
| Macromolecule | Name: SAGA / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Recombinant expression: No |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.8 MDa |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL |
|---|---|
| Buffer | pH: 8 / Details: 500 mM NaCl, 30 mM Hepes, 2 mM EGTA |
| Staining | Type: NEGATIVE Details: Grids with adsorbed protein stained with 2% w/v uranyl acetate |
| Grid | Details: 300 mesh Cu/Rh grid with glow discharge |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Date | Oct 10, 2012 |
| Image recording | Category: CCD / Film or detector model: GATAN ULTRASCAN 1000 (2k x 2k) / Average electron dose: 20 e/Å2 / Bits/pixel: 16 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 66038 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 1.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | IMAGIC |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 25.8 Å / Resolution method: OTHER / Software - Name: IMAGIC / Number images used: 18916 |
| Final two d classification | Number classes: 500 |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)