[English] 日本語
 Yorodumi
Yorodumi- EMDB-26180: Calcitonin Receptor in complex with Gs and rat amylin peptide, CT... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Calcitonin Receptor in complex with Gs and rat amylin peptide, CT-like state | ||||||||||||||||||||||||
 Map data Map data | post-processed consensus map | ||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
 Keywords Keywords | Amylin receptor / GPCR / calcitonin / CT-like / MEMBRANE PROTEIN | ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationCalcitonin-like ligand receptors / calcitonin binding / calcitonin family receptor activity / amylin receptor complex 1 / amylin receptor complex 2 / calcitonin family receptor signaling pathway / amylin receptor complex 3 / amylin receptor activity / calcitonin receptor activity / calcitonin gene-related peptide receptor signaling pathway ...Calcitonin-like ligand receptors / calcitonin binding / calcitonin family receptor activity / amylin receptor complex 1 / amylin receptor complex 2 / calcitonin family receptor signaling pathway / amylin receptor complex 3 / amylin receptor activity / calcitonin receptor activity / calcitonin gene-related peptide receptor signaling pathway / calcitonin gene-related peptide receptor activity / amylin receptor 3 signaling pathway / amylin receptor 2 signaling pathway / amylin receptor 1 signaling pathway / amylin receptor signaling pathway / Calcitonin-like ligand receptors / negative regulation of ossification / negative regulation of bone resorption / eating behavior / negative regulation of osteoclast differentiation / response to amyloid-beta / adenylate cyclase-activating G protein-coupled bile acid receptor signaling pathway / adenylate cyclase-activating serotonin receptor signaling pathway / positive regulation of cAMP/PKA signal transduction / regulation of skeletal muscle contraction / PKA activation in glucagon signalling / hair follicle placode formation / developmental growth / intracellular transport / bone resorption / D1 dopamine receptor binding / vascular endothelial cell response to laminar fluid shear stress / renal water homeostasis / activation of adenylate cyclase activity / Hedgehog 'off' state / cellular response to acidic pH / adenylate cyclase-activating adrenergic receptor signaling pathway / regulation of mRNA stability / inclusion body / cellular response to glucagon stimulus / positive regulation of calcium-mediated signaling / osteoclast differentiation / sensory perception of pain / secretory granule / intracellular glucose homeostasis / ossification / response to glucocorticoid / acrosomal vesicle / positive regulation of insulin secretion involved in cellular response to glucose stimulus / adenylate cyclase activator activity / trans-Golgi network membrane / negative regulation of inflammatory response to antigenic stimulus / response to prostaglandin E / hormone activity / protein homooligomerization / bone development / platelet aggregation / protein destabilization / cognition / glucose metabolic process / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / positive regulation of insulin secretion / G protein-coupled acetylcholine receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / sensory perception of smell / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / G beta:gamma signalling through BTK / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / positive regulation of cold-induced thermogenesis / heterotrimeric G-protein complex / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / amyloid-beta binding / G-protein beta-subunit binding / extracellular vesicle Similarity search - Function | ||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /   | ||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||||||||||||||
 Authors Authors | Cao J / Belousoff MJ / Johnson RM / Wootten DL / Sexton PM | ||||||||||||||||||||||||
| Funding support |  Australia, Australia,  Japan, 7 items Japan, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: A structural basis for amylin receptor phenotype. Authors: Jianjun Cao / Matthew J Belousoff / Yi-Lynn Liang / Rachel M Johnson / Tracy M Josephs / Madeleine M Fletcher / Arthur Christopoulos / Debbie L Hay / Radostin Danev / Denise Wootten / Patrick M Sexton /    Abstract: Amylin receptors (AMYRs) are heterodimers of the calcitonin (CT) receptor (CTR) and one of three receptor activity-modifying proteins (RAMPs), AMYR, AMYR, and AMYR. Selective AMYR agonists and dual ...Amylin receptors (AMYRs) are heterodimers of the calcitonin (CT) receptor (CTR) and one of three receptor activity-modifying proteins (RAMPs), AMYR, AMYR, and AMYR. Selective AMYR agonists and dual AMYR/CTR agonists are being developed as obesity treatments; however, the molecular basis for peptide binding and selectivity is unknown. We determined the structure and dynamics of active AMYRs with amylin, AMYR with salmon CT (sCT), AMYR with sCT or human CT (hCT), and CTR with amylin, sCT, or hCT. The conformation of amylin-bound complexes was similar for all AMYRs, constrained by the RAMP, and an ordered midpeptide motif that we call the bypass motif. The CT-bound AMYR complexes were distinct, overlapping the CT-bound CTR complexes. Our findings indicate that activation of AMYRs by CT-based peptides is distinct from their activation by amylin-based peptides. This has important implications for the development of AMYR therapeutics. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26180.map.gz emd_26180.map.gz | 85.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26180-v30.xml emd-26180-v30.xml emd-26180.xml emd-26180.xml | 40.7 KB 40.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26180_fsc.xml emd_26180_fsc.xml | 10.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_26180.png emd_26180.png | 29.7 KB | ||
| Masks |  emd_26180_msk_1.map emd_26180_msk_1.map | 91.1 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-26180.cif.gz emd-26180.cif.gz | 8.3 KB | ||
| Others |  emd_26180_additional_1.map.gz emd_26180_additional_1.map.gz emd_26180_additional_2.map.gz emd_26180_additional_2.map.gz emd_26180_additional_3.map.gz emd_26180_additional_3.map.gz emd_26180_additional_4.map.gz emd_26180_additional_4.map.gz emd_26180_additional_5.map.gz emd_26180_additional_5.map.gz emd_26180_additional_6.map.gz emd_26180_additional_6.map.gz emd_26180_half_map_1.map.gz emd_26180_half_map_1.map.gz emd_26180_half_map_2.map.gz emd_26180_half_map_2.map.gz | 82.8 MB 84.6 MB 43.8 MB 43.4 MB 44.8 MB 85 MB 84.5 MB 84.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26180 http://ftp.pdbj.org/pub/emdb/structures/EMD-26180 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26180 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26180 | HTTPS FTP |
-Related structure data
| Related structure data |  7tyiMC  7tyfC  7tyhC  7tylC  7tynC  7tyoC  7tywC  7tyxC  7tyyC  7tzfC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26180.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26180.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | post-processed consensus map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.856 Å | ||||||||||||||||||||||||||||||||||||
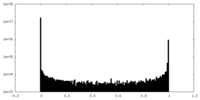
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_26180_msk_1.map emd_26180_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
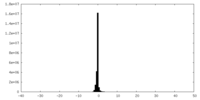
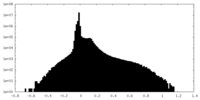
| Density Histograms |
-Additional map: Composite map from overlaying locallly refined maps
| File | emd_26180_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite map from overlaying locallly refined maps | ||||||||||||
| Projections & Slices |
| ||||||||||||
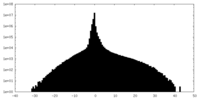
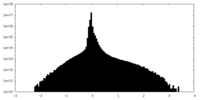
| Density Histograms |
-Additional map: ECD focussed map, post-processed
| File | emd_26180_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | ECD focussed map, post-processed | ||||||||||||
| Projections & Slices |
| ||||||||||||
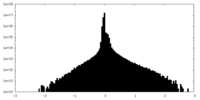
| Density Histograms |
-Additional map: ECD focussed map, unfiltered
| File | emd_26180_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | ECD focussed map, unfiltered | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: unsharpened consensus map
| File | emd_26180_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | unsharpened consensus map | ||||||||||||
| Projections & Slices |
| ||||||||||||
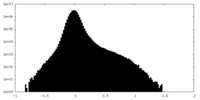
| Density Histograms |
-Additional map: receptor focussed map, unfiltered
| File | emd_26180_additional_5.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | receptor focussed map, unfiltered | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: receptor focussed map, post-processed
| File | emd_26180_additional_6.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | receptor focussed map, post-processed | ||||||||||||
| Projections & Slices |
| ||||||||||||
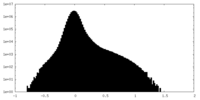
| Density Histograms |
-Half map: half map 1
| File | emd_26180_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map 2
| File | emd_26180_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Calcitonin Receptor in complex with Gs and rat amylin peptide, CT...
| Entire | Name: Calcitonin Receptor in complex with Gs and rat amylin peptide, CT-like state |
|---|---|
| Components |
|
-Supramolecule #1: Calcitonin Receptor in complex with Gs and rat amylin peptide, CT...
| Supramolecule | Name: Calcitonin Receptor in complex with Gs and rat amylin peptide, CT-like state type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 |
|---|
-Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 45.685406 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKNTIVKQM RILHVNGFNG EGGEEDPQAA RSNSDGEKA TKVQDIKNNL KEAIETIVAA MSNLVPPVEL ANPENQFRVD YILSVMNVPD FDFPPEFYEH AKALWEDEGV R ACYERSNE ...String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKNTIVKQM RILHVNGFNG EGGEEDPQAA RSNSDGEKA TKVQDIKNNL KEAIETIVAA MSNLVPPVEL ANPENQFRVD YILSVMNVPD FDFPPEFYEH AKALWEDEGV R ACYERSNE YQLIDCAQYF LDKIDVIKQA DYVPSDQDLL RCRVLTSGIF ETKFQVDKVN FHMFDVGGQR DERRKWIQCF ND VTAIIFV VASSSYNMVI REDNQTNRLQ AALKLFDSIW NNKWLRDTSV ILFLNKQDLL AEKVLAGKSK IEDYFPEFAR YTT PEDATP EPGEDPRVTR AKYFIRDEFL RISTASGDGR HYCYPHFTCS VDTENIRRVF NDCRDIIQRM HLRQYELL UniProtKB: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.534062 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV ...String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV TSSGDTTCAL WDIETGQQTT TFTGHTGDVM SLSLAPDTRL FVSGACDASA KLWDVREGMC RQTFTGHESD IN AICFFPN GNAFATGSDD ATCRLFDLRA DQELMTYSHD NIICGITSVS FSKSGRLLLA GYDDFNCNVW DALKADRAGV LAG HDNRVS CLGVTDDGMA VATGSWDSFL KIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: Nanobody 35
| Macromolecule | Name: Nanobody 35 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 15.140742 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTVSSH HHHHHEPEA |
-Macromolecule #5: Islet amyloid polypeptide
| Macromolecule | Name: Islet amyloid polypeptide / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 3.924426 KDa |
| Sequence | String: KCNTATCATQ RLANFLVRSS NNLGPVLPPT NVGSNTY(NH2) UniProtKB: Islet amyloid polypeptide |
-Macromolecule #6: Calcitonin receptor
| Macromolecule | Name: Calcitonin receptor / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58.469594 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDLEVLFQG PAAFSNQTYP TIEPKPFLYV VGRKKMMDAQ YKCYDRMQQL PAYQGEGPYC NRTWDGWLC WDDTPAGVLS YQFCPDYFPD FDPSEKVTKY CDEKGVWFKH PENNRTWSNY TMCNAFTPEK LKNAYVLYYL A IVGHSLSI ...String: MKTIIALSYI FCLVFADYKD DDDLEVLFQG PAAFSNQTYP TIEPKPFLYV VGRKKMMDAQ YKCYDRMQQL PAYQGEGPYC NRTWDGWLC WDDTPAGVLS YQFCPDYFPD FDPSEKVTKY CDEKGVWFKH PENNRTWSNY TMCNAFTPEK LKNAYVLYYL A IVGHSLSI FTLVISLGIF VFFRSLGCQR VTLHKNMFLT YILNSMIIII HLVEVVPNGE LVRRDPVSCK ILHFFHQYMM AC NYFWMLC EGIYLHTLIV VAVFTEKQRL RWYYLLGWGF PLVPTTIHAI TRAVYFNDNC WLSVETHLLY IIHGPVMAAL VVN FFFLLN IVRVLVTKMR ETHEAESHMY LKAVKATMIL VPLLGIQFVV FPWRPSNKML GKIYDYVMHS LIHFQGFFVA TIYC FCNNE VQTTVKRQWA QFKIQWNQRW GRRPSNRSAR AAAAAAEAGD IPIYICHQEL RNEPANNQGE ESAEIIPLNI IEQES SAPA GLEVLFQGPH HHHHHHH UniProtKB: Calcitonin receptor |
-Macromolecule #7: PALMITIC ACID
| Macromolecule | Name: PALMITIC ACID / type: ligand / ID: 7 / Number of copies: 10 / Formula: PLM |
|---|---|
| Molecular weight | Theoretical: 256.424 Da |
| Chemical component information |  ChemComp-PLM: |
-Macromolecule #8: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 8 / Number of copies: 2 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 13 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
 Movie
Movie Controller
Controller































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)