[English] 日本語
 Yorodumi
Yorodumi- EMDB-25142: Human STING bound to both cGAMP and 1-[(2-chloro-6-fluorophenyl)m... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-25142 | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human STING bound to both cGAMP and 1-[(2-chloro-6-fluorophenyl)methyl]-3,3-dimethyl-2-oxo-N-[(2,4,6-trifluorophenyl)methyl]-2,3-dihydro-1H-indole-6-carboxamide (Compound 53) | |||||||||||||||||||||
 Map data Map data | ||||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||
 Keywords Keywords | Sting / agonist / activate conformation / oligomer / IMMUNE SYSTEM | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationSTING complex / STAT6-mediated induction of chemokines / protein localization to endoplasmic reticulum / 2',3'-cyclic GMP-AMP binding / cyclic-di-GMP binding / STING mediated induction of host immune responses / serine/threonine protein kinase complex / positive regulation of type I interferon-mediated signaling pathway / IRF3-mediated induction of type I IFN / cGAS/STING signaling pathway ...STING complex / STAT6-mediated induction of chemokines / protein localization to endoplasmic reticulum / 2',3'-cyclic GMP-AMP binding / cyclic-di-GMP binding / STING mediated induction of host immune responses / serine/threonine protein kinase complex / positive regulation of type I interferon-mediated signaling pathway / IRF3-mediated induction of type I IFN / cGAS/STING signaling pathway / proton channel activity / pattern recognition receptor signaling pathway / reticulophagy / cytoplasmic pattern recognition receptor signaling pathway / cellular response to exogenous dsRNA / protein complex oligomerization / autophagosome membrane / positive regulation of macroautophagy / autophagosome assembly / positive regulation of type I interferon production / positive regulation of DNA-binding transcription factor activity / cellular response to interferon-beta / positive regulation of defense response to virus by host / signaling adaptor activity / antiviral innate immune response / endoplasmic reticulum-Golgi intermediate compartment membrane / activation of innate immune response / Regulation of innate immune responses to cytosolic DNA / protein serine/threonine kinase binding / positive regulation of interferon-beta production / autophagosome / cytoplasmic vesicle membrane / secretory granule membrane / SARS-CoV-1 activates/modulates innate immune responses / peroxisome / regulation of inflammatory response / defense response to virus / DNA-binding transcription factor binding / RNA polymerase II-specific DNA-binding transcription factor binding / mitochondrial outer membrane / endosome / cilium / ciliary basal body / Golgi membrane / innate immune response / ubiquitin protein ligase binding / Neutrophil degranulation / endoplasmic reticulum membrane / protein kinase binding / perinuclear region of cytoplasm / SARS-CoV-2 activates/modulates innate and adaptive immune responses / protein homodimerization activity / positive regulation of transcription by RNA polymerase II / nucleoplasm / identical protein binding / plasma membrane / cytosol Similarity search - Function | |||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||
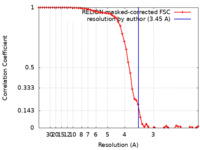
| Method | single particle reconstruction / cryo EM / Resolution: 3.45 Å | |||||||||||||||||||||
 Authors Authors | Lu D / Shang G | |||||||||||||||||||||
| Funding support |  United States, 6 items United States, 6 items
| |||||||||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Activation of STING by targeting a pocket in the transmembrane domain. Authors: Defen Lu / Guijun Shang / Jie Li / Yong Lu / Xiao-Chen Bai / Xuewu Zhang /  Abstract: Stimulator of interferon genes (STING) is an adaptor protein in innate immunity against DNA viruses or bacteria. STING-mediated immunity could be exploited in the development of vaccines or cancer ...Stimulator of interferon genes (STING) is an adaptor protein in innate immunity against DNA viruses or bacteria. STING-mediated immunity could be exploited in the development of vaccines or cancer immunotherapies. STING is a transmembrane dimeric protein that is located in the endoplasmic reticulum or in the Golgi apparatus. STING is activated by the binding of its cytoplasmic ligand-binding domain to cyclic dinucleotides that are produced by the DNA sensor cyclic GMP-AMP (cGAMP) synthase or by invading bacteria. Cyclic dinucleotides induce a conformational change in the STING ligand-binding domain, which leads to a high-order oligomerization of STING that is essential for triggering the downstream signalling pathways. However, the cGAMP-induced STING oligomers tend to dissociate in solution and have not been resolved to high resolution, which limits our understanding of the activation mechanism. Here we show that a small-molecule agonist, compound 53 (C53), promotes the oligomerization and activation of human STING through a mechanism orthogonal to that of cGAMP. We determined a cryo-electron microscopy structure of STING bound to both C53 and cGAMP, revealing a stable oligomer that is formed by side-by-side packing and has a curled overall shape. Notably, C53 binds to a cryptic pocket in the STING transmembrane domain, between the two subunits of the STING dimer. This binding triggers outward shifts of transmembrane helices in the dimer, and induces inter-dimer interactions between these helices to mediate the formation of the high-order oligomer. Our functional analyses show that cGAMP and C53 together induce stronger activation of STING than either ligand alone. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25142.map.gz emd_25142.map.gz | 14.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25142-v30.xml emd-25142-v30.xml emd-25142.xml emd-25142.xml | 11.8 KB 11.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25142_fsc.xml emd_25142_fsc.xml | 5.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_25142.png emd_25142.png | 120 KB | ||
| Filedesc metadata |  emd-25142.cif.gz emd-25142.cif.gz | 5.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25142 http://ftp.pdbj.org/pub/emdb/structures/EMD-25142 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25142 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25142 | HTTPS FTP |
-Validation report
| Summary document |  emd_25142_validation.pdf.gz emd_25142_validation.pdf.gz | 551.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_25142_full_validation.pdf.gz emd_25142_full_validation.pdf.gz | 551.5 KB | Display | |
| Data in XML |  emd_25142_validation.xml.gz emd_25142_validation.xml.gz | 8.6 KB | Display | |
| Data in CIF |  emd_25142_validation.cif.gz emd_25142_validation.cif.gz | 10.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25142 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25142 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25142 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25142 | HTTPS FTP |
-Related structure data
| Related structure data |  7siiMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_25142.map.gz / Format: CCP4 / Size: 15.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25142.map.gz / Format: CCP4 / Size: 15.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
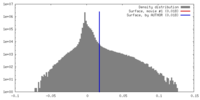
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : human STING
| Entire | Name: human STING |
|---|---|
| Components |
|
-Supramolecule #1: human STING
| Supramolecule | Name: human STING / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Stimulator of interferon genes protein
| Macromolecule | Name: Stimulator of interferon genes protein / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.553398 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MPHSSLHPSI PCPRGHGAQK AALVLLSACL VTLWGLGEPP EHTLRYLVLH LASLQLGLLL NGVCSLAEEL RHIHSRYRGS YWRTVRACL GCPLRRGALL LLSIYFYYSL PNAVGPPFTW MLALLGLSQA LNILLGLKGL APAEISAVCE KGNFNVAHGL A WSYYIGYL ...String: MPHSSLHPSI PCPRGHGAQK AALVLLSACL VTLWGLGEPP EHTLRYLVLH LASLQLGLLL NGVCSLAEEL RHIHSRYRGS YWRTVRACL GCPLRRGALL LLSIYFYYSL PNAVGPPFTW MLALLGLSQA LNILLGLKGL APAEISAVCE KGNFNVAHGL A WSYYIGYL RLILPELQAR IRTYNQHYNN LLRGAVSQRL YILLPLDCGV PDNLSMADPN IRFLDKLPQQ TGDRAGIKDR VY SNSIYEL LENGQRAGTC VLEYATPLQT LFAMSQYSQA GFSREDRLEQ AKLFCRTLED ILADAPESQN NCRLIAYQEP ADD SSFSLS QEVLRHLRQE EKEEVTVGTS SGLEVLFQ UniProtKB: Stimulator of interferon genes protein |
-Macromolecule #2: 1-[(2-chloro-6-fluorophenyl)methyl]-3,3-dimethyl-2-oxo-N-[(2,4,6-...
| Macromolecule | Name: 1-[(2-chloro-6-fluorophenyl)methyl]-3,3-dimethyl-2-oxo-N-[(2,4,6-trifluorophenyl)methyl]-2,3-dihydro-1H-indole-6-carboxamide type: ligand / ID: 2 / Number of copies: 2 / Formula: 9IM |
|---|---|
| Molecular weight | Theoretical: 490.877 Da |
| Chemical component information | 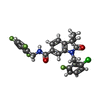 ChemComp-9IM: |
-Macromolecule #3: cGAMP
| Macromolecule | Name: cGAMP / type: ligand / ID: 3 / Number of copies: 2 / Formula: 1SY |
|---|---|
| Molecular weight | Theoretical: 674.411 Da |
| Chemical component information | 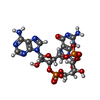 ChemComp-1SY: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: NITROGEN |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-7sii: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)