+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-2514 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | ER membrane-associated ribosome from HeLa cells after treatment with non-silencing control siRNA | |||||||||
 マップデータ マップデータ | ER membrane-associated ribosome from HeLa cells after treatment with non-silencing control siRNA | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | endoplasmic reticulum / translocon / ribosome / oligosaccharyltransferase / OST / protein-conducting channel / Sec61 / translocon-associated protein complex / TRAP | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | サブトモグラム平均法 / クライオ電子顕微鏡法 / 解像度: 40.0 Å | |||||||||
 データ登録者 データ登録者 | Pfeffer S / Dudek J / Gogala M / Schorr S / Linxweiler J / Lang S / Becker T / Beckmann R / Zimmermann R / Foerster F | |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2014 ジャーナル: Nat Commun / 年: 2014タイトル: Structure of the mammalian oligosaccharyl-transferase complex in the native ER protein translocon. 著者: Stefan Pfeffer / Johanna Dudek / Marko Gogala / Stefan Schorr / Johannes Linxweiler / Sven Lang / Thomas Becker / Roland Beckmann / Richard Zimmermann / Friedrich Förster /  要旨: In mammalian cells, proteins are typically translocated across the endoplasmic reticulum (ER) membrane in a co-translational mode by the ER protein translocon, comprising the protein-conducting ...In mammalian cells, proteins are typically translocated across the endoplasmic reticulum (ER) membrane in a co-translational mode by the ER protein translocon, comprising the protein-conducting channel Sec61 and additional complexes involved in nascent chain processing and translocation. As an integral component of the translocon, the oligosaccharyl-transferase complex (OST) catalyses co-translational N-glycosylation, one of the most common protein modifications in eukaryotic cells. Here we use cryoelectron tomography, cryoelectron microscopy single-particle analysis and small interfering RNA-mediated gene silencing to determine the overall structure, oligomeric state and position of OST in the native ER protein translocon of mammalian cells in unprecedented detail. The observed positioning of OST in close proximity to Sec61 provides a basis for understanding how protein translocation into the ER and glycosylation of nascent proteins are structurally coupled. The overall spatial organization of the native translocon, as determined here, serves as a reliable framework for further hypothesis-driven studies. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_2514.map.gz emd_2514.map.gz | 28.3 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-2514-v30.xml emd-2514-v30.xml emd-2514.xml emd-2514.xml | 9.6 KB 9.6 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_2514.tif emd_2514.tif | 816.3 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2514 http://ftp.pdbj.org/pub/emdb/structures/EMD-2514 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2514 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2514 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_2514.map.gz / 形式: CCP4 / 大きさ: 29.8 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_2514.map.gz / 形式: CCP4 / 大きさ: 29.8 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | ER membrane-associated ribosome from HeLa cells after treatment with non-silencing control siRNA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 2.88 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
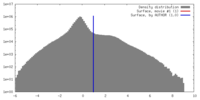
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : ER membrane-associated ribosome from HeLa cells after treatment w...
| 全体 | 名称: ER membrane-associated ribosome from HeLa cells after treatment with non-silencing control siRNA |
|---|---|
| 要素 |
|
-超分子 #1000: ER membrane-associated ribosome from HeLa cells after treatment w...
| 超分子 | 名称: ER membrane-associated ribosome from HeLa cells after treatment with non-silencing control siRNA タイプ: sample / ID: 1000 / Number unique components: 2 |
|---|
-超分子 #1: ER membrane-associated 80S ribosome
| 超分子 | 名称: ER membrane-associated 80S ribosome / タイプ: complex / ID: 1 / 組換発現: No / Ribosome-details: ribosome-eukaryote: ALL |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 株: HeLa / 別称: Human / 細胞中の位置: Endoplasmic reticulum Homo sapiens (ヒト) / 株: HeLa / 別称: Human / 細胞中の位置: Endoplasmic reticulum |
-分子 #1: ER protein translocon
| 分子 | 名称: ER protein translocon / タイプ: protein_or_peptide / ID: 1 / 組換発現: No |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 株: HeLa / 別称: Human / 細胞中の位置: Endoplasmic reticulum Homo sapiens (ヒト) / 株: HeLa / 別称: Human / 細胞中の位置: Endoplasmic reticulum |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | サブトモグラム平均法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 2 mg/mL |
|---|---|
| グリッド | 詳細: Lacey carbon molybdenum grid |
| 凍結 | 凍結剤: ETHANE-PROPANE MIXTURE / チャンバー内湿度: 70 % / 装置: FEI VITROBOT MARK IV / 手法: Blot 3 seconds before plunging. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 日付 | 2012年10月31日 |
| 撮影 | カテゴリ: CCD フィルム・検出器のモデル: FEI FALCON I (4k x 4k) 平均電子線量: 60 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 8.0 µm / 最小 デフォーカス(公称値): 7.0 µm / 倍率(公称値): 29000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER Tilt series - Axis1 - Min angle: -60 ° / Tilt series - Axis1 - Max angle: 60 ° |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 詳細 | Candidate particles were localized using template matching and cross correlation peaks in areas containing rough ER were visually inspected to identify true positive matches. At the selected coordinates, unbinned subtomograms were reconstructed individually from the weighted back-projections and aligned to a template. |
|---|---|
| 最終 再構成 | 想定した対称性 - 点群: C1 (非対称) / 解像度のタイプ: BY AUTHOR / 解像度: 40.0 Å / 解像度の算出法: OTHER / ソフトウェア - 名称: av3, tom, PyTom / 使用したサブトモグラム数: 295 |
| CTF補正 | 詳細: projection |
 ムービー
ムービー コントローラー
コントローラー













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)