[English] 日本語
 Yorodumi
Yorodumi- EMDB-2346: Bacteriophage phi6 procapsids with different composition of acces... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2346 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Bacteriophage phi6 procapsids with different composition of accessory proteins | |||||||||
 Map data Map data | recombinant phi6 procapsid composed of P1 and P4 subunits (i.e. without the P2 and P7 subunits) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | dsRNA virus / Cystoviridae / procapsid / RNA polymerase / P7 subunit / location / occupancy | |||||||||
| Biological species |  Pseudomonas phage phi6 (virus) Pseudomonas phage phi6 (virus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 12.4 Å | |||||||||
 Authors Authors | Nemecek D / Qiao J / Mindich L / Steven AC / Heymann JB | |||||||||
 Citation Citation |  Journal: J Virol / Year: 2012 Journal: J Virol / Year: 2012Title: Packaging accessory protein P7 and polymerase P2 have mutually occluding binding sites inside the bacteriophage 6 procapsid. Authors: Daniel Nemecek / Jian Qiao / Leonard Mindich / Alasdair C Steven / J Bernard Heymann /  Abstract: Bacteriophage 6 is a double-stranded RNA (dsRNA) virus whose genome is packaged sequentially as three single-stranded RNA (ssRNA) segments into an icosahedral procapsid which serves as a compartment ...Bacteriophage 6 is a double-stranded RNA (dsRNA) virus whose genome is packaged sequentially as three single-stranded RNA (ssRNA) segments into an icosahedral procapsid which serves as a compartment for genome replication and transcription. The procapsid shell consists of 60 copies each of P1(A) and P1(B), two nonequivalent conformers of the P1 protein. Hexamers of the packaging ATPase P4 are mounted over the 5-fold vertices, and monomers of the RNA-dependent RNA polymerase (P2) attach to the inner surface, near the 3-fold axes. A fourth protein, P7, is needed for packaging and also promotes assembly. We used cryo-electron microscopy to localize P7 by difference mapping of procapsids with different protein compositions. We found that P7 resides on the interior surface of the P1 shell and appears to be monomeric. Its binding sites are arranged around the 3-fold axes, straddling the interface between two P1(A) subunits. Thus, P7 may promote assembly by stabilizing an initiation complex. Only about 20% of the 60 P7 binding sites were occupied in our preparations. P7 density overlaps P2 density similarly mapped, implying mutual occlusion. The known structure of the 12 homolog fits snugly into the P7 density. Both termini-which have been implicated in RNA binding-are oriented toward the adjacent 5-fold vertex, the entry pathway of ssRNA segments. Thus, P7 may promote packaging either by interacting directly with incoming RNA or by modulating the structure of the translocation pore. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2346.map.gz emd_2346.map.gz | 227.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2346-v30.xml emd-2346-v30.xml emd-2346.xml emd-2346.xml | 9.4 KB 9.4 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD-2346.png EMD-2346.png | 2.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2346 http://ftp.pdbj.org/pub/emdb/structures/EMD-2346 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2346 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2346 | HTTPS FTP |
-Validation report
| Summary document |  emd_2346_validation.pdf.gz emd_2346_validation.pdf.gz | 298.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2346_full_validation.pdf.gz emd_2346_full_validation.pdf.gz | 297.4 KB | Display | |
| Data in XML |  emd_2346_validation.xml.gz emd_2346_validation.xml.gz | 7.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2346 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2346 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2346 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2346 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2346.map.gz / Format: CCP4 / Size: 238.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2346.map.gz / Format: CCP4 / Size: 238.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | recombinant phi6 procapsid composed of P1 and P4 subunits (i.e. without the P2 and P7 subunits) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.63 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
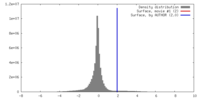
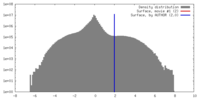
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : recombinant phi6 procapsid composed of P1 and P4 subunits
| Entire | Name: recombinant phi6 procapsid composed of P1 and P4 subunits |
|---|---|
| Components |
|
-Supramolecule #1000: recombinant phi6 procapsid composed of P1 and P4 subunits
| Supramolecule | Name: recombinant phi6 procapsid composed of P1 and P4 subunits type: sample / ID: 1000 Oligomeric state: icosahedral shell without acessory proteins Number unique components: 3 |
|---|---|
| Molecular weight | Theoretical: 12.6 MDa |
-Supramolecule #1: Pseudomonas phage phi6
| Supramolecule | Name: Pseudomonas phage phi6 / type: virus / ID: 1 / Name.synonym: bacteriophage phi6 / NCBI-ID: 10879 / Sci species name: Pseudomonas phage phi6 / Virus type: VIRION / Virus isolate: SPECIES / Virus enveloped: No / Virus empty: Yes / Syn species name: bacteriophage phi6 |
|---|---|
| Host (natural) | Organism:  Pseudomonas syringae (bacteria) / synonym: BACTERIA(EUBACTERIA) Pseudomonas syringae (bacteria) / synonym: BACTERIA(EUBACTERIA) |
| Host system | Organism:  |
| Molecular weight | Theoretical: 12.6 MDa |
| Virus shell | Shell ID: 1 / Diameter: 450 Å / T number (triangulation number): 2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 10 mg/mL |
|---|---|
| Buffer | pH: 8 / Details: 10mM Tris, 5mM MgCl2 |
| Grid | Details: Q-foil 2/2/300 mesh copper grid |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 80 % / Chamber temperature: 120 K / Instrument: FEI VITROBOT MARK I / Method: Blot for 2 seconds before plunging |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200FEG |
|---|---|
| Date | Feb 20, 2011 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: NIKON SUPER COOLSCAN 9000 / Digitization - Sampling interval: 6.35 µm / Number real images: 4047 / Average electron dose: 15 e/Å2 / Bits/pixel: 16 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 38800 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.05 µm / Nominal defocus min: 0.65 µm / Nominal magnification: 38000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN |
- Image processing
Image processing
| Details | particles were selected manually, CTF was determined from whole micrographs |
|---|---|
| CTF correction | Details: CTF was determined from whole micrographs |
| Final reconstruction | Applied symmetry - Point group: I (icosahedral) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 12.4 Å / Resolution method: FSC 0.33 CUT-OFF / Software - Name: Bsoft / Number images used: 4047 |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)