[English] 日本語
 Yorodumi
Yorodumi- EMDB-2195: Characterization of the insertase for beta-barrel proteins of the... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2195 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Characterization of the insertase for beta-barrel proteins of the outer mitochondrial membrane. 3-D reconstruction of the TOB complex | |||||||||
 Map data Map data | 3D reconstruction of TOB complexes isolated using the 9xHis tag on the Tob37 subunit. The TOB complex is a hetero trimer containing one copy each of Tob37, Tob38 and Tob37. Total mol. Wgt. Is 140 kDa. In this sample there are three isoforms of Tob55 54.7, 54.1 and 50.7 kDa respectively. This affects the reconstruction. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TOB/SAM complex / beta-barrel proteins / Tob55/Sam50 / mitochondria outer membrane / cryo-electron microscopy | |||||||||
| Biological species |  Neurospora crassa (fungus) Neurospora crassa (fungus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 14.7 Å | |||||||||
 Authors Authors | Klein A / Israel L / Lackey SWK / Nargang FE / Imhof A / Baumeister W / Neupert W / Thomas DR | |||||||||
 Citation Citation |  Journal: J Cell Biol / Year: 2012 Journal: J Cell Biol / Year: 2012Title: Characterization of the insertase for β-barrel proteins of the outer mitochondrial membrane. Authors: Astrid Klein / Lars Israel / Sebastian W K Lackey / Frank E Nargang / Axel Imhof / Wolfgang Baumeister / Walter Neupert / Dennis R Thomas /  Abstract: The TOB-SAM complex is an essential component of the mitochondrial outer membrane that mediates the insertion of β-barrel precursor proteins into the membrane. We report here its isolation and ...The TOB-SAM complex is an essential component of the mitochondrial outer membrane that mediates the insertion of β-barrel precursor proteins into the membrane. We report here its isolation and determine its size, composition, and structural organization. The complex from Neurospora crassa was composed of Tob55-Sam50, Tob38-Sam35, and Tob37-Sam37 in a stoichiometry of 1:1:1 and had a molecular mass of 140 kD. A very minor fraction of the purified complex was associated with one Mdm10 protein. Using molecular homology modeling for Tob55 and cryoelectron microscopy reconstructions of the TOB complex, we present a model of the TOB-SAM complex that integrates biochemical and structural data. We discuss our results and the structural model in the context of a possible mechanism of the TOB insertase. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2195.map.gz emd_2195.map.gz | 9.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2195-v30.xml emd-2195-v30.xml emd-2195.xml emd-2195.xml | 12.6 KB 12.6 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD-2195.jpg EMD-2195.jpg | 22.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2195 http://ftp.pdbj.org/pub/emdb/structures/EMD-2195 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2195 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2195 | HTTPS FTP |
-Related structure data
| Related structure data | 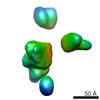 2196C  2197C  2200C  2201C  2202C  2203C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2195.map.gz / Format: CCP4 / Size: 10.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2195.map.gz / Format: CCP4 / Size: 10.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 3D reconstruction of TOB complexes isolated using the 9xHis tag on the Tob37 subunit. The TOB complex is a hetero trimer containing one copy each of Tob37, Tob38 and Tob37. Total mol. Wgt. Is 140 kDa. In this sample there are three isoforms of Tob55 54.7, 54.1 and 50.7 kDa respectively. This affects the reconstruction. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.78 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : TOB complex mitochondrial outer membrane complex required for ins...
| Entire | Name: TOB complex mitochondrial outer membrane complex required for inserting beta-barrel proteins into the outer membrane. |
|---|---|
| Components |
|
-Supramolecule #1000: TOB complex mitochondrial outer membrane complex required for ins...
| Supramolecule | Name: TOB complex mitochondrial outer membrane complex required for inserting beta-barrel proteins into the outer membrane. type: sample / ID: 1000 / Details: The complexes were monodisperse. Oligomeric state: heterotrimer with one subunit each Tob37,Tob38 and Tob55 Number unique components: 3 |
|---|---|
| Molecular weight | Experimental: 140 KDa / Theoretical: 140 KDa Method: Blue native gel electrophoresis and Isotope dilution mass spectroscopy analysis of bands isolated from BNGE gels. |
-Macromolecule #1: Tob37
| Macromolecule | Name: Tob37 / type: protein_or_peptide / ID: 1 Details: The TOB complex contains one copy each of T0b37, Tob38 and Tob55 Number of copies: 1 / Oligomeric state: monmer / Recombinant expression: No |
|---|---|
| Source (natural) | Organism:  Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane |
| Molecular weight | Experimental: 48.6 KDa / Theoretical: 48.6 KDa |
-Macromolecule #2: Tob38
| Macromolecule | Name: Tob38 / type: protein_or_peptide / ID: 2 Details: The TOB complex contains one copy each of T0b37, Tob38 and Tob55 Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: No |
|---|---|
| Source (natural) | Organism:  Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane |
| Molecular weight | Experimental: 37.3 KDa / Theoretical: 37.3 KDa |
-Macromolecule #3: Tob55
| Macromolecule | Name: Tob55 / type: protein_or_peptide / ID: 3 Details: The TOB complex contains one copy each of T0b37, Tob38 and Tob55 Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: No |
|---|---|
| Source (natural) | Organism:  Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane Neurospora crassa (fungus) / Strain: Tob37HT / Organelle: mitochondria / Location in cell: outer membrane |
| Molecular weight | Experimental: 54.7 KDa / Theoretical: 54.7 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 8.5 Details: 1mM PMSF, 0.08%(v/v) Triton X-100, 50 mM HEPES pH 8.5 |
| Grid | Details: lacey carbon films on 200 mesh Molybdenum grids |
| Vitrification | Cryogen name: ETHANE / Chamber temperature: 120 K / Instrument: HOMEMADE PLUNGER Method: blot for 4-5 seconds before plunging with whatman filter paper |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Temperature | Max: 95 K |
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected using the live FFT at imaging magnification. |
| Date | Oct 11, 2010 |
| Image recording | Category: CCD / Film or detector model: FEI EAGLE (4k x 4k) / Number real images: 546 / Average electron dose: 20 e/Å2 Details: Images collected using TOM_acquisition software. 546 good micrographs were CTF corrected for phase. 175,000 particles were automatically selected. In the end 76700 were included in the final reconstruction. |
| Tilt angle min | 0 |
| Tilt angle max | 0 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 84270 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: -3.7 µm / Nominal defocus min: -0.5 µm / Nominal magnification: 62000 |
| Sample stage | Specimen holder: Gatan 656 side entry holder / Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| CTF correction | Details: Phase and astigmatism correction applied to each micrograph |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 14.7 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: SPIDER, TOM_toolbox Details: Final maps were reconstructed from images that had stable alignment parameters over the last 4 rounds of refinement. Stable was defined by absolute accumulated changes in theta and psi of ...Details: Final maps were reconstructed from images that had stable alignment parameters over the last 4 rounds of refinement. Stable was defined by absolute accumulated changes in theta and psi of the projection matched of less than 10 degrees. Number images used: 1 |
 Movie
Movie Controller
Controller



 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















