[English] 日本語
 Yorodumi
Yorodumi- EMDB-21037: Structure of NPC1-like intracellular cholesterol transporter 1 (N... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21037 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of NPC1-like intracellular cholesterol transporter 1 (NPC1L1) in complex with an ezetimibe analog | |||||||||
 Map data Map data | EM map of NPC1L1-EzePS | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cholesterol / Transporter / Tunnel / Inhibitor / Ezetimibe / LIPID TRANSPORT-INHIBITOR complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationIntestinal lipid absorption / cellular response to sterol depletion / vitamin E binding / vitamin E metabolic process / vitamin transport / cholesterol import / sterol transport / intestinal cholesterol absorption / lipid carrier activity / lipoprotein metabolic process ...Intestinal lipid absorption / cellular response to sterol depletion / vitamin E binding / vitamin E metabolic process / vitamin transport / cholesterol import / sterol transport / intestinal cholesterol absorption / lipid carrier activity / lipoprotein metabolic process / myosin V binding / cholesterol transport / heterocyclic compound binding / cholesterol binding / cholesterol biosynthetic process / response to muscle activity / cholesterol homeostasis / brush border membrane / small GTPase binding / apical plasma membrane / response to xenobiotic stimulus / protein homodimerization activity / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.5 Å | |||||||||
 Authors Authors | Huang CS / Yu X | |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2020 Journal: Sci Adv / Year: 2020Title: Cryo-EM structures of NPC1L1 reveal mechanisms of cholesterol transport and ezetimibe inhibition. Authors: Ching-Shin Huang / Xinchao Yu / Preston Fordstrom / Kaylee Choi / Ben C Chung / Soung-Hun Roh / Wah Chiu / Mingyue Zhou / Xiaoshan Min / Zhulun Wang /  Abstract: The intestinal absorption of cholesterol is mediated by a multipass membrane protein, Niemann-Pick C1-Like 1 (NPC1L1), the molecular target of a cholesterol lowering therapy ezetimibe. While ...The intestinal absorption of cholesterol is mediated by a multipass membrane protein, Niemann-Pick C1-Like 1 (NPC1L1), the molecular target of a cholesterol lowering therapy ezetimibe. While ezetimibe gained Food and Drug Administration approval in 2002, its mechanism of action has remained unclear. Here, we present two cryo-electron microscopy structures of NPC1L1, one in its apo form and the other complexed with ezetimibe. The apo form represents an open state in which the N-terminal domain (NTD) interacts loosely with the rest of NPC1L1, leaving the NTD central cavity accessible for cholesterol loading. The ezetimibe-bound form signifies a closed state in which the NTD rotates ~60°, creating a continuous tunnel enabling cholesterol movement into the plasma membrane. Ezetimibe blocks cholesterol transport by occluding the tunnel instead of competing with cholesterol binding. These findings provide insight into the molecular mechanisms of NPC1L1-mediated cholesterol transport and ezetimibe inhibition, paving the way for more effective therapeutic development. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21037.map.gz emd_21037.map.gz | 59.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21037-v30.xml emd-21037-v30.xml emd-21037.xml emd-21037.xml | 12.8 KB 12.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_21037.png emd_21037.png | 48.5 KB | ||
| Filedesc metadata |  emd-21037.cif.gz emd-21037.cif.gz | 6.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21037 http://ftp.pdbj.org/pub/emdb/structures/EMD-21037 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21037 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21037 | HTTPS FTP |
-Related structure data
| Related structure data |  6v3hMC  6v3fC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_21037.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21037.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM map of NPC1L1-EzePS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.05878 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : NPC1-like intracellular cholesterol transporter 1 (NPC1L1) in com...
| Entire | Name: NPC1-like intracellular cholesterol transporter 1 (NPC1L1) in complex with an inhibitor |
|---|---|
| Components |
|
-Supramolecule #1: NPC1-like intracellular cholesterol transporter 1 (NPC1L1) in com...
| Supramolecule | Name: NPC1-like intracellular cholesterol transporter 1 (NPC1L1) in complex with an inhibitor type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: NPC1-like intracellular cholesterol transporter 1
| Macromolecule | Name: NPC1-like intracellular cholesterol transporter 1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 147.640188 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: ELYTPKHEAG VCTFYEECGK NPELSGGLTS LSNVSCLSNT PARHVTGEHL ALLQRICPRL YNGPNTTFAC CSTKQLLSLE SSMSITKAL LTRCPACSDN FVSLHCHNTC SPDQSLFINV TRVVERGAGE PPAVVAYEAF YQRSFAEKAY ESCSQVRIPA A ASLAVGSM ...String: ELYTPKHEAG VCTFYEECGK NPELSGGLTS LSNVSCLSNT PARHVTGEHL ALLQRICPRL YNGPNTTFAC CSTKQLLSLE SSMSITKAL LTRCPACSDN FVSLHCHNTC SPDQSLFINV TRVVERGAGE PPAVVAYEAF YQRSFAEKAY ESCSQVRIPA A ASLAVGSM CGVYGSALCN AQRWLNFQGD TGNGLAPLDI TFHLLEPGQA LPDGIQPLNG KIAPCNESQG DDSAVCSCQD CA ASCPVIP PPEALRPSFY MGRMPGWLAL IIIFTAVFVL LSAVLVRLRV VSNRNKNKAE GPQEAPKLPH KHKLSPHTIL GRF FQNWGT RVASWPLTVL ALSFIVVIAL AAGLTFIELT TDPVELWSAP KSQARKEKSF HDEHFGPFFR TNQIFVTARN RSSY KYDSL LLGSKNFSGI LSLDFLLELL ELQERLRHLQ VWSPEAERNI SLQDICYAPL NPYNTSLSDC CVNSLLQYFQ NNRTL LMLT ANQTLNGQTS LVDWKDHFLY CANAPLTFKD GTSLALSCMA DYGAPVFPFL AVGGYQGTDY SEAEALIITF SLNNYP ADD PRMAQAKLWE EAFLKEMESF QRNTSDKFQV AFSAERSLED EINRTTIQDL PVFAVSYIIV FLYISLALGS YSRCSRV AV ESKATLGLGG VIVVLGAVLA AMGFYSYLGV PSSLVIIQVV PFLVLAVGAD NIFIFVLEYQ RLPRMPGEQR EAHIGRTL G SVAPSMLLCS LSEAICFFLG ALTPMPAVRT FALTSGLAII LDFLLQMTAF VALLSLDSKR QEASRPDVLC CFSTRKLPP PKEKEGLLLR FFRKIYAPFL LHRFIRPVVM LLFLTLFGAN LYLMCNINVG LDQELALPKD SYLIDYFLFL NRYLEVGPPV YFVTTSGFN FSSEAGMNAT CSSAGCKSFS LTQKIQYASE FPDQSYVAIA ASSWVDDFID WLTPSSSCCR LYIRGPHKDE F CPSTDTSF NCLKNCMNRT LGPVRPTAEQ FHKYLPWFLN DPPNIRCPKG GLAAYRTSVN LSSDGQVIAS QFMAYHKPLR NS QDFTEAL RASRLLAANI TADLRKVPGT DPNFEVFPYT ISNVFYQQYL TVLPEGIFTL ALCFVPTFVV CYLLLGLDMC SGI LNLLSI IMILVDTIGL MAVWGISYNA VSLINLVTAV GMSVEFVSHI TRSFAVSTKP TRLERAKDAT VFMGSAVFAG VAMT NFPGI LILGFAQAQL IQIFFFRLNL LITLLGLLHG LVFLPVVLSY LGPDVNQALV QEEKLASEAA VAPEPSCPQY PSPAD ADAN VNYGFAPELA HGANAARSSL PKSDQKFENL YFQGDYKDDD DKHHHHHHHH HH UniProtKB: NPC1-like intracellular cholesterol transporter 1 |
-Macromolecule #3: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 3 / Number of copies: 6 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #4: CHOLESTEROL HEMISUCCINATE
| Macromolecule | Name: CHOLESTEROL HEMISUCCINATE / type: ligand / ID: 4 / Number of copies: 2 / Formula: Y01 |
|---|---|
| Molecular weight | Theoretical: 486.726 Da |
| Chemical component information |  ChemComp-Y01: |
-Macromolecule #5: 4-[(2S,3R)-3-[(3S)-3-(4-fluorophenyl)-3-hydroxypropyl]-1-(4-{3-[(...
| Macromolecule | Name: 4-[(2S,3R)-3-[(3S)-3-(4-fluorophenyl)-3-hydroxypropyl]-1-(4-{3-[(methylsulfonyl)amino]prop-1-yn-1-yl}phenyl)-4-oxoazetidin-2-yl]phenyl beta-D-glucopyranosiduronic acid type: ligand / ID: 5 / Number of copies: 1 / Formula: QO1 |
|---|---|
| Molecular weight | Theoretical: 698.712 Da |
| Chemical component information | 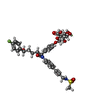 ChemComp-QO1: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 5 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 48.61 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.5 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.0) / Number images used: 252262 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.0) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.0) |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















