[English] 日本語
 Yorodumi
Yorodumi- EMDB-21014: Cryo-EM reconstruction of the thermophilic bacteriophage P74-26 s... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21014 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM reconstruction of the thermophilic bacteriophage P74-26 small terminase- asymmetric II | |||||||||
 Map data Map data | An asymmetric cryoEM reconstruction of the small terminase protein from the thermophilic bacteriophage P74-26, asymmetric II | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  Thermus virus P74-26 Thermus virus P74-26 | |||||||||
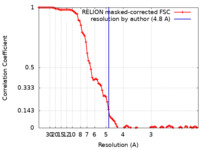
| Method | single particle reconstruction / cryo EM / Resolution: 4.8 Å | |||||||||
 Authors Authors | Hayes JA / Brendan BJ / Gaubitz C / Stone NP / Kelch BA | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2020 Journal: J Biol Chem / Year: 2020Title: A thermophilic phage uses a small terminase protein with a fixed helix-turn-helix geometry. Authors: Janelle A Hayes / Brendan J Hilbert / Christl Gaubitz / Nicholas P Stone / Brian A Kelch /  Abstract: Tailed bacteriophages use a DNA-packaging motor to encapsulate their genome during viral particle assembly. The small terminase (TerS) component of this DNA-packaging machinery acts as a molecular ...Tailed bacteriophages use a DNA-packaging motor to encapsulate their genome during viral particle assembly. The small terminase (TerS) component of this DNA-packaging machinery acts as a molecular matchmaker that recognizes both the viral genome and the main motor component, the large terminase (TerL). However, how TerS binds DNA and the TerL protein remains unclear. Here we identified gp83 of the thermophilic bacteriophage P74-26 as the TerS protein. We found that TerS oligomerizes into a nonamer that binds DNA, stimulates TerL ATPase activity, and inhibits TerL nuclease activity. A cryo-EM structure of TerS revealed that it forms a ring with a wide central pore and radially arrayed helix-turn-helix domains. The structure further showed that these helix-turn-helix domains, which are thought to bind DNA by wrapping the double helix around the ring, are rigidly held in an orientation distinct from that seen in other TerS proteins. This rigid arrangement of the putative DNA-binding domain imposed strong constraints on how TerS can bind DNA. Finally, the TerS structure lacked the conserved C-terminal β-barrel domain used by other TerS proteins for binding TerL. This suggests that a well-ordered C-terminal β-barrel domain is not required for TerS to carry out its matchmaking function. Our work highlights a thermophilic system for studying the role of small terminase proteins in viral maturation and presents the structure of TerS, revealing key differences between this thermophilic phage and its mesophilic counterparts. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21014.map.gz emd_21014.map.gz | 5.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21014-v30.xml emd-21014-v30.xml emd-21014.xml emd-21014.xml | 18 KB 18 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_21014_fsc.xml emd_21014_fsc.xml | 9.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_21014.png emd_21014.png | 144.5 KB | ||
| Others |  emd_21014_half_map_1.map.gz emd_21014_half_map_1.map.gz emd_21014_half_map_2.map.gz emd_21014_half_map_2.map.gz | 49.8 MB 49.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21014 http://ftp.pdbj.org/pub/emdb/structures/EMD-21014 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21014 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21014 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_21014.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21014.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | An asymmetric cryoEM reconstruction of the small terminase protein from the thermophilic bacteriophage P74-26, asymmetric II | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.059 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: Half map 2 of an asymmetric cryoEM reconstruction...
| File | emd_21014_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 of an asymmetric cryoEM reconstruction of the small terminase protein from the thermophilic bacteriophage P74-26, asymmetric II | ||||||||||||
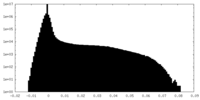
| Projections & Slices |
| ||||||||||||
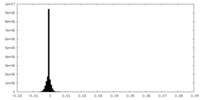
| Density Histograms |
-Half map: Half map 1 of an asymmetric cryoEM reconstruction...
| File | emd_21014_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 of an asymmetric cryoEM reconstruction of the small terminase protein from the thermophilic bacteriophage P74-26, asymmetric II | ||||||||||||
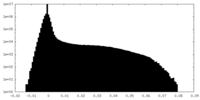
| Projections & Slices |
| ||||||||||||
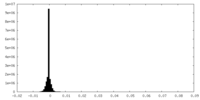
| Density Histograms |
- Sample components
Sample components
-Entire : Gene product 83 of the Thermus thermophilus bacteriophage P74-26
| Entire | Name: Gene product 83 of the Thermus thermophilus bacteriophage P74-26 |
|---|---|
| Components |
|
-Supramolecule #1: Gene product 83 of the Thermus thermophilus bacteriophage P74-26
| Supramolecule | Name: Gene product 83 of the Thermus thermophilus bacteriophage P74-26 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Thermus virus P74-26 Thermus virus P74-26 |
| Recombinant expression | Organism:  |
| Molecular weight | Experimental: 171 KDa |
-Macromolecule #1: Small terminase protein of Thermus thermophilus bacteriophage P74...
| Macromolecule | Name: Small terminase protein of Thermus thermophilus bacteriophage P74-26, asymmetric II type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Thermus virus P74-26 Thermus virus P74-26 |
| Recombinant expression | Organism:  |
| Sequence | String: GPHMSVSFRD RVLKLYLLGF DPSEIAQTLS LDAKRKVTEE EVLHVLAEAR ELLSALPSLE DIRAEVGQAL ERARIFQKDL LAIYQNMLRN YNAMMEGLTE HPDGTPVIGV RPADIAAMAD RIMKIDQERI TALLNSLKVL GHVGSTTAGA LPSATELVSV EELVAEVADE TPKT |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.4 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||
| Grid | Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: OTHER / Details: 20 mA | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV | ||||||||||||
| Details | This sample was monodisperse (PDI= 1.000) |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Number grids imaged: 2 / Number real images: 2822 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.6 µm / Calibrated defocus min: 1.4 µm / Calibrated magnification: 13000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)