[English] 日本語
 Yorodumi
Yorodumi- EMDB-20878: Allosteric coupling between alpha-rings of the 20S proteasome, 20... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-20878 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Allosteric coupling between alpha-rings of the 20S proteasome, 20S singly capped with a PA26/V230F | |||||||||
 Map data Map data | Allosteric coupling between alpha-rings of the 20S proteasome, 20S singly capped with a PA26/V230F | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | proteasome / PA26/V230F / singly-capped / HYDROLASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationproteasome activator complex / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / regulation of proteasomal protein catabolic process / proteasomal protein catabolic process / endopeptidase activity / ubiquitin-dependent protein catabolic process / proteolysis / cytoplasm Similarity search - Function | |||||||||
| Biological species |   Thermoplasma acidophilum (acidophilic) / Thermoplasma acidophilum (acidophilic) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Yu Z / Cheng Y | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2020 Journal: Nat Commun / Year: 2020Title: Allosteric coupling between α-rings of the 20S proteasome. Authors: Zanlin Yu / Yadong Yu / Feng Wang / Alexander G Myasnikov / Philip Coffino / Yifan Cheng /  Abstract: Proteasomal machinery performs essential regulated protein degradation in eukaryotes. Classic proteasomes are symmetric, with a regulatory ATPase docked at each end of the cylindrical 20S. Asymmetric ...Proteasomal machinery performs essential regulated protein degradation in eukaryotes. Classic proteasomes are symmetric, with a regulatory ATPase docked at each end of the cylindrical 20S. Asymmetric complexes are also present in cells, either with a single ATPase or with an ATPase and non-ATPase at two opposite ends. The mechanism that populates these different proteasomal complexes is unknown. Using archaea homologs, we construct asymmetric forms of proteasomes. We demonstrate that the gate conformation of the two opposite ends of 20S are coupled: binding one ATPase opens a gate locally, and also opens the opposite gate allosterically. Such allosteric coupling leads to cooperative binding of proteasomal ATPases to 20S and promotes formation of proteasomes symmetrically configured with two identical ATPases. It may also promote formation of asymmetric complexes with an ATPase and a non-ATPase at opposite ends. We propose that in eukaryotes a similar mechanism regulates the composition of the proteasomal population. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_20878.map.gz emd_20878.map.gz | 117.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-20878-v30.xml emd-20878-v30.xml emd-20878.xml emd-20878.xml | 20.2 KB 20.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_20878.png emd_20878.png | 69.6 KB | ||
| Filedesc metadata |  emd-20878.cif.gz emd-20878.cif.gz | 5.6 KB | ||
| Others |  emd_20878_additional.map.gz emd_20878_additional.map.gz emd_20878_half_map_1.map.gz emd_20878_half_map_1.map.gz emd_20878_half_map_2.map.gz emd_20878_half_map_2.map.gz | 62.9 MB 115.9 MB 115.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-20878 http://ftp.pdbj.org/pub/emdb/structures/EMD-20878 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20878 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20878 | HTTPS FTP |
-Related structure data
| Related structure data |  6utgMC  6utfC  6uthC  6utiC  6utjC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_20878.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_20878.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Allosteric coupling between alpha-rings of the 20S proteasome, 20S singly capped with a PA26/V230F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.234 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: #1
| File | emd_20878_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
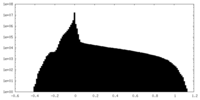
| Density Histograms |
-Half map: half map 1
| File | emd_20878_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
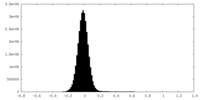
| Density Histograms |
-Half map: half map 2
| File | emd_20878_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
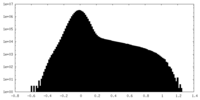
| Density Histograms |
- Sample components
Sample components
-Entire : The complex of Archaea 20S singly capped by one PA26/V230F
| Entire | Name: The complex of Archaea 20S singly capped by one PA26/V230F |
|---|---|
| Components |
|
-Supramolecule #1: The complex of Archaea 20S singly capped by one PA26/V230F
| Supramolecule | Name: The complex of Archaea 20S singly capped by one PA26/V230F type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
-Macromolecule #1: Proteasome subunit beta
| Macromolecule | Name: Proteasome subunit beta / type: protein_or_peptide / ID: 1 / Number of copies: 14 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
| Molecular weight | Theoretical: 22.294848 KDa |
| Recombinant expression | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Sequence | String: TTTVGITLKD AVIMATERRV TMENFIMHKN GKKLFQIDTY TGMTIAGLVG DAQVLVRYMK AELELYRLQR RVNMPIEAVA TLLSNMLNQ VKYMPYMVQL LVGGIDTAPH VFSIDAAGGS VEDIYASTGS GSPFVYGVLE SQYSEKMTVD EGVDLVIRAI S AAKQRDSA ...String: TTTVGITLKD AVIMATERRV TMENFIMHKN GKKLFQIDTY TGMTIAGLVG DAQVLVRYMK AELELYRLQR RVNMPIEAVA TLLSNMLNQ VKYMPYMVQL LVGGIDTAPH VFSIDAAGGS VEDIYASTGS GSPFVYGVLE SQYSEKMTVD EGVDLVIRAI S AAKQRDSA SGGMIDVAVI TRKDGYVQLP TDQIESRIRK LGLIL UniProtKB: Proteasome subunit beta |
-Macromolecule #2: Proteasome subunit alpha
| Macromolecule | Name: Proteasome subunit alpha / type: protein_or_peptide / ID: 2 / Number of copies: 7 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
| Molecular weight | Theoretical: 25.125619 KDa |
| Recombinant expression | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Sequence | String: AYDRAITVFS PDGRLFQVEY AREAVKKGST ALGMKFANGV LLISDKKVRS RLIEQNSIEK IQLIDDYVAA VTSGLVADAR VLVDFARIS AQQEKVTYGS LVNIENLVKR VADQMQQYTQ YGGVRPYGVS LIFAGIDQIG PRLFDCDPAG TINEYKATAI G SGKDAVVS ...String: AYDRAITVFS PDGRLFQVEY AREAVKKGST ALGMKFANGV LLISDKKVRS RLIEQNSIEK IQLIDDYVAA VTSGLVADAR VLVDFARIS AQQEKVTYGS LVNIENLVKR VADQMQQYTQ YGGVRPYGVS LIFAGIDQIG PRLFDCDPAG TINEYKATAI G SGKDAVVS FLEREYKENL PEKEAVTLGI KALKSSLEEG EELKAPEIAS ITVGNKYRIY DQEEVKKFL UniProtKB: Proteasome subunit alpha |
-Macromolecule #3: Proteasome activator protein PA26
| Macromolecule | Name: Proteasome activator protein PA26 / type: protein_or_peptide / ID: 3 / Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 24.995404 KDa |
| Recombinant expression | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Sequence | String: KRAALIQNLR DSYTETSSFA VIEEWAAGTL QEIEGIAKAA AEAHGVIRNS TYGRAQAEKS PEQLLGVLQR YQDLCHNVYC QAETIRTVI AIRIPEHKEE DNLGVAVQHA VLKIIDELEI KTLGSGEKSG SGGAPTPIGM YALREYLSAR STVEDKLLGS V DAESGKTK ...String: KRAALIQNLR DSYTETSSFA VIEEWAAGTL QEIEGIAKAA AEAHGVIRNS TYGRAQAEKS PEQLLGVLQR YQDLCHNVYC QAETIRTVI AIRIPEHKEE DNLGVAVQHA VLKIIDELEI KTLGSGEKSG SGGAPTPIGM YALREYLSAR STVEDKLLGS V DAESGKTK GGSQSPSLLL ELRQIDADFM LKVELATTHL STMVRAVINA YLLNWKKLIQ PRTGTDHMFS UniProtKB: Proteasome activator protein PA26 |
-Macromolecule #4: Proteasome subunit alpha
| Macromolecule | Name: Proteasome subunit alpha / type: protein_or_peptide / ID: 4 / Number of copies: 7 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
| Molecular weight | Theoretical: 24.718178 KDa |
| Recombinant expression | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Sequence | String: RAITVFSPDG RLFQVEYARE AVKKGSTALG MKFANGVLLI SDKKVRSRLI EQNSIEAIQL IDDYVAAVTS GLVADARVLV DFARISAQQ EKVTYGSLVN IENLVKRVAD QMQQYTQYGG VRPYGVSLIF AGIDQIGPRL FDCDPAGTIN EYKATAIGSG K DAVVSFLE ...String: RAITVFSPDG RLFQVEYARE AVKKGSTALG MKFANGVLLI SDKKVRSRLI EQNSIEAIQL IDDYVAAVTS GLVADARVLV DFARISAQQ EKVTYGSLVN IENLVKRVAD QMQQYTQYGG VRPYGVSLIF AGIDQIGPRL FDCDPAGTIN EYKATAIGSG K DAVVSFLE REYKENLPEK EAVTLGIKAL KSSLEEGEEL KAPEIASITV GNKYRIYDQE EVKKFL UniProtKB: Proteasome subunit alpha |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Details: unspecified |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 50000 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)