[English] 日本語
 Yorodumi
Yorodumi- EMDB-19841: HSV-1 DNA polymerase-processivity factor complex in exonuclease s... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | HSV-1 DNA polymerase-processivity factor complex in exonuclease state with 1-bp DNA mismatch processivity factor focused refinement | |||||||||
 Map data Map data | Local refinement map from cryoSPARC | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA / Polymerase / Complex / TRANSFERASE | |||||||||
| Biological species |   Human alphaherpesvirus 1 strain KOS / synthetic construct (others) Human alphaherpesvirus 1 strain KOS / synthetic construct (others) | |||||||||
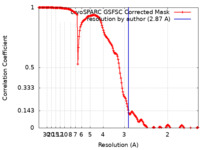
| Method | single particle reconstruction / cryo EM / Resolution: 2.87 Å | |||||||||
 Authors Authors | Gustavsson E / Grunewald K / Elias P / Hallberg BM | |||||||||
| Funding support |  Sweden, Sweden,  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2024 Journal: Nucleic Acids Res / Year: 2024Title: Dynamics of the Herpes simplex virus DNA polymerase holoenzyme during DNA synthesis and proof-reading revealed by Cryo-EM. Authors: Emil Gustavsson / Kay Grünewald / Per Elias / B Martin Hällberg /   Abstract: Herpes simplex virus 1 (HSV-1), a double-stranded DNA virus, replicates using seven essential proteins encoded by its genome. Among these, the UL30 DNA polymerase, complexed with the UL42 ...Herpes simplex virus 1 (HSV-1), a double-stranded DNA virus, replicates using seven essential proteins encoded by its genome. Among these, the UL30 DNA polymerase, complexed with the UL42 processivity factor, orchestrates leading and lagging strand replication of the 152 kb viral genome. UL30 polymerase is a prime target for antiviral therapy, and resistance to current drugs can arise in immunocompromised individuals. Using electron cryo-microscopy (cryo-EM), we unveil the dynamic changes of the UL30/UL42 complex with DNA in three distinct states. First, a pre-translocation state with an open fingers domain ready for nucleotide incorporation. Second, a halted elongation state where the fingers close, trapping dATP in the dNTP pocket. Third, a DNA-editing state involving significant conformational changes to allow DNA realignment for exonuclease activity. Additionally, the flexible UL30 C-terminal domain interacts with UL42, forming an extended positively charged surface binding to DNA, thereby enhancing processive synthesis. These findings highlight substantial structural shifts in the polymerase and its DNA interactions during replication, offering insights for future antiviral drug development. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_19841.map.gz emd_19841.map.gz | 211 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-19841-v30.xml emd-19841-v30.xml emd-19841.xml emd-19841.xml | 21.3 KB 21.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_19841_fsc.xml emd_19841_fsc.xml | 15.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_19841.png emd_19841.png | 54.9 KB | ||
| Masks |  emd_19841_msk_1.map emd_19841_msk_1.map | 421.9 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-19841.cif.gz emd-19841.cif.gz | 5.7 KB | ||
| Others |  emd_19841_additional_1.map.gz emd_19841_additional_1.map.gz emd_19841_half_map_1.map.gz emd_19841_half_map_1.map.gz emd_19841_half_map_2.map.gz emd_19841_half_map_2.map.gz | 2 MB 391.3 MB 391.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-19841 http://ftp.pdbj.org/pub/emdb/structures/EMD-19841 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19841 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19841 | HTTPS FTP |
-Validation report
| Summary document |  emd_19841_validation.pdf.gz emd_19841_validation.pdf.gz | 1.2 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_19841_full_validation.pdf.gz emd_19841_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  emd_19841_validation.xml.gz emd_19841_validation.xml.gz | 25.3 KB | Display | |
| Data in CIF |  emd_19841_validation.cif.gz emd_19841_validation.cif.gz | 32.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19841 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19841 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19841 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19841 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_19841.map.gz / Format: CCP4 / Size: 421.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_19841.map.gz / Format: CCP4 / Size: 421.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Local refinement map from cryoSPARC | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_19841_msk_1.map emd_19841_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
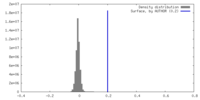
| Density Histograms |
-Additional map: Locally filtered local refinement map from cryoSPARC
| File | emd_19841_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Locally filtered local refinement map from cryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
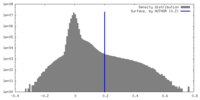
| Density Histograms |
-Half map: Halfmap A of local refinement map from cryoSPARC
| File | emd_19841_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Halfmap A of local refinement map from cryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
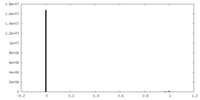
| Density Histograms |
-Half map: Halfmap B of local refinement map from cryoSPARC
| File | emd_19841_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Halfmap B of local refinement map from cryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
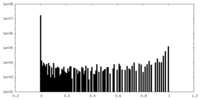
| Density Histograms |
- Sample components
Sample components
-Entire : HSV-1 DNA polymerase-processivity factor complex in exonuclease s...
| Entire | Name: HSV-1 DNA polymerase-processivity factor complex in exonuclease state with 1-bp DNA mismatch |
|---|---|
| Components |
|
-Supramolecule #1: HSV-1 DNA polymerase-processivity factor complex in exonuclease s...
| Supramolecule | Name: HSV-1 DNA polymerase-processivity factor complex in exonuclease state with 1-bp DNA mismatch type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Supramolecule #2: DNA polymerase-processivity factor
| Supramolecule | Name: DNA polymerase-processivity factor / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Human alphaherpesvirus 1 strain KOS Human alphaherpesvirus 1 strain KOS |
-Supramolecule #3: DNA
| Supramolecule | Name: DNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Macromolecule #1: DNA polymerase processivity factor
| Macromolecule | Name: DNA polymerase processivity factor / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human alphaherpesvirus 1 strain KOS Human alphaherpesvirus 1 strain KOS |
| Recombinant expression | Organism:  |
| Sequence | String: MTDSPGGVAP ASPVEDASDA SLGQPEEGAP CQVVLQGAEL NGILQAFAPL RTSLLDSLLV MGDRGILIHN TIFGEQVFLP LEHSQFSRY RWRGPTAAFL SLVDQKRSLL SVFRANQYPD LRRVELAITG QAPFRTLVQR IWTTTSDGEA VELASETLMK R ELTSFVVL ...String: MTDSPGGVAP ASPVEDASDA SLGQPEEGAP CQVVLQGAEL NGILQAFAPL RTSLLDSLLV MGDRGILIHN TIFGEQVFLP LEHSQFSRY RWRGPTAAFL SLVDQKRSLL SVFRANQYPD LRRVELAITG QAPFRTLVQR IWTTTSDGEA VELASETLMK R ELTSFVVL VPQGTPDVQL RLTRPQLTKV LNATGADSAT PTTFELGVNG KFSVFTTSTC VTFAAREEGV SSSTSTQVQI LS NALTKAG QAAANAKTVY GENTHRTFSV VVDDCSMRAV LRRLQVGGGT LKFFLTTPVP SLCVTATGPN AVSAVFLLKP QKI CLDWLG HSQGSPSAGS SASRASGSEP TDSQDSASDA VSHGDPEDLD GAARAGEAGA LHACPMPSST TRVTPTTKRG RSGG EDARA DTALKKPKTG SPTAPPPADP VPLDTEDDSD AADGTAARPA APDARSGSRY ACYFRDLPTG EASPGAFSAF RGGPQ TPYG FGFP |
-Macromolecule #2: Primer DNA
| Macromolecule | Name: Primer DNA / type: dna / ID: 2 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Sequence | String: GCCACTACGA CACCTTGATC GCCTCGCAGC CGTCCAACCA ACTC(DRG) |
-Macromolecule #3: Template DNA
| Macromolecule | Name: Template DNA / type: dna / ID: 3 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Sequence | String: ATTTGCTGAC CTTTGTTCTG GGTGAGTTGG TTGGACGGCT GCGAGGCGAT CAAGGTGTCG TAGTGGC |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.8 Component:
Details: 20mM HEPES pH7.8, 150mM NaCl, 5mM CaCl2, 2mM DTT | |||||||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY | |||||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 95 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 58.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.3 µm / Nominal magnification: 165000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|
 Movie
Movie Controller
Controller
































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)