+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | DtpB hexamer from Streptomyces lividans | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Heme / iron / dye-type peroxidase / METAL BINDING PROTEIN / OXIDOREDUCTASE | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Streptomyces lividans (bacteria) Streptomyces lividans (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.02 Å | |||||||||
 Authors Authors | Worrall JAR / Chaplin AK / Allport T | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Protein Sci / Year: 2024 Journal: Protein Sci / Year: 2024Title: The oligomeric states of dye-decolorizing peroxidases from Streptomyces lividans and their implications for mechanism of substrate oxidation. Authors: Marina Lučić / Thomas Allport / Thomas A Clarke / Lewis J Williams / Michael T Wilson / Amanda K Chaplin / Jonathan A R Worrall /  Abstract: A common evolutionary mechanism in biology to drive function is protein oligomerization. In prokaryotes, the symmetrical assembly of repeating protein units to form homomers is widespread, yet ...A common evolutionary mechanism in biology to drive function is protein oligomerization. In prokaryotes, the symmetrical assembly of repeating protein units to form homomers is widespread, yet consideration in vitro of whether such assemblies have functional or mechanistic consequences is often overlooked. Dye-decolorizing peroxidases (DyPs) are one such example, where their dimeric α + β barrel units can form various oligomeric states, but the oligomer influence, if any, on mechanism and function has received little attention. In this work, we have explored the oligomeric state of three DyPs found in Streptomyces lividans, each with very different mechanistic behaviors in their reactions with hydrogen peroxide and organic substrates. Using analytical ultracentrifugation, we reveal that except for one of the A-type DyPs where only a single sedimenting species is detected, oligomer states ranging from homodimers to dodecamers are prevalent in solution. Using cryo-EM on preparations of the B-type DyP, we determined a 3.02 Å resolution structure of a hexamer assembly that corresponds to the dominant oligomeric state in solution as determined by analytical ultracentrifugation. Furthermore, cryo-EM data detected sub-populations of higher-order oligomers, with one of these formed by an arrangement of two B-type DyP hexamers to give a dodecamer assembly. Our solution and structural insights of these oligomer states provide a new framework to consider previous mechanistic studies of these DyP members and are discussed in terms of long-range electron transfer for substrate oxidation and in the "storage" of oxidizable equivalents on the heme until a two-electron donor is available. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_19568.map.gz emd_19568.map.gz | 266.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-19568-v30.xml emd-19568-v30.xml emd-19568.xml emd-19568.xml | 14.5 KB 14.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_19568_fsc.xml emd_19568_fsc.xml | 23.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_19568.png emd_19568.png | 95.8 KB | ||
| Filedesc metadata |  emd-19568.cif.gz emd-19568.cif.gz | 5.4 KB | ||
| Others |  emd_19568_half_map_1.map.gz emd_19568_half_map_1.map.gz emd_19568_half_map_2.map.gz emd_19568_half_map_2.map.gz | 498.1 MB 498.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-19568 http://ftp.pdbj.org/pub/emdb/structures/EMD-19568 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19568 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19568 | HTTPS FTP |
-Related structure data
| Related structure data |  8rwyMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_19568.map.gz / Format: CCP4 / Size: 536.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_19568.map.gz / Format: CCP4 / Size: 536.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.652 Å | ||||||||||||||||||||||||||||||||||||
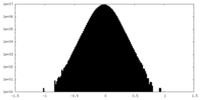
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_19568_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
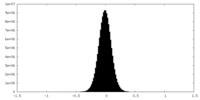
| Density Histograms |
-Half map: #2
| File | emd_19568_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
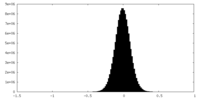
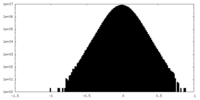
| Density Histograms |
- Sample components
Sample components
-Entire : Hexamer complex of Dye-type peroxidase B from Streptomyces Lividans
| Entire | Name: Hexamer complex of Dye-type peroxidase B from Streptomyces Lividans |
|---|---|
| Components |
|
-Supramolecule #1: Hexamer complex of Dye-type peroxidase B from Streptomyces Lividans
| Supramolecule | Name: Hexamer complex of Dye-type peroxidase B from Streptomyces Lividans type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Streptomyces lividans (bacteria) Streptomyces lividans (bacteria) |
-Macromolecule #1: Dyp-type peroxidase family
| Macromolecule | Name: Dyp-type peroxidase family / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Streptomyces lividans (bacteria) Streptomyces lividans (bacteria) |
| Molecular weight | Theoretical: 34.172223 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGGEVEEPEP QMVLSPLTSA AIFLVVTIDS GGEDTVRDLL SDVASLERAV GFRAQPDGRL SCVTGIGSEA WDRLFSGARP AGLHPFREL DGPVHRAVAT PGDLLFHIRA SRLDLCFALA TEIMGRLRGA VTPQDEVHGF KYFDERDMLG FVDGTENPTG A AARRAVLV ...String: MGGEVEEPEP QMVLSPLTSA AIFLVVTIDS GGEDTVRDLL SDVASLERAV GFRAQPDGRL SCVTGIGSEA WDRLFSGARP AGLHPFREL DGPVHRAVAT PGDLLFHIRA SRLDLCFALA TEIMGRLRGA VTPQDEVHGF KYFDERDMLG FVDGTENPTG A AARRAVLV GAEDPAFAGG SYAVVQKYLH DIDAWEGLSV EAQERVIGRR KMTDVELSDD VKPADSHVAL TSVTGPDGSD LE ILRDNMP FGSVGREEFG TYFIGYARTP EVTETMLERM FLGTASAPHD RILDFSTAVT GSLFFTPAAD FLEDLSARP UniProtKB: Dyp-type peroxidase family |
-Macromolecule #2: PROTOPORPHYRIN IX CONTAINING FE
| Macromolecule | Name: PROTOPORPHYRIN IX CONTAINING FE / type: ligand / ID: 2 / Number of copies: 6 / Formula: HEM |
|---|---|
| Molecular weight | Theoretical: 616.487 Da |
| Chemical component information |  ChemComp-HEM: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Grid | Material: COPPER / Mesh: 300 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 47.19 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.7000000000000001 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)