[English] 日本語
 Yorodumi
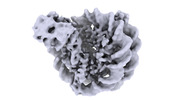
Yorodumi- EMDB-18497: Human Sirtuin 6 in complex with nucleosome - structure showing H2... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human Sirtuin 6 in complex with nucleosome - structure showing H2A tail path | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Histone deacetylase / Sirtuin / nucleosome / TRANSCRIPTION | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.78 Å | |||||||||
 Authors Authors | Smirnova E / Bignon E / Schultz P / Papai G / Ben-Shem A | |||||||||
| Funding support |  France, 1 items France, 1 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2024 Journal: Elife / Year: 2024Title: Binding to nucleosome poises human SIRT6 for histone H3 deacetylation. Authors: Ekaterina Smirnova / Emmanuelle Bignon / Patrick Schultz / Gabor Papai / Adam Ben Shem /  Abstract: Sirtuin 6 (SIRT6) is an NAD-dependent histone H3 deacetylase that is prominently found associated with chromatin, attenuates transcriptionally active promoters and regulates DNA repair, metabolic ...Sirtuin 6 (SIRT6) is an NAD-dependent histone H3 deacetylase that is prominently found associated with chromatin, attenuates transcriptionally active promoters and regulates DNA repair, metabolic homeostasis and lifespan. Unlike other sirtuins, it has low affinity to free histone tails but demonstrates strong binding to nucleosomes. It is poorly understood how SIRT6 docking on nucleosomes stimulates its histone deacetylation activity. Here, we present the structure of human SIRT6 bound to a nucleosome determined by cryogenic electron microscopy. The zinc finger domain of SIRT6 associates tightly with the acidic patch of the nucleosome through multiple arginine anchors. The Rossmann fold domain binds to the terminus of the looser DNA half of the nucleosome, detaching two turns of the DNA from the histone octamer and placing the NAD binding pocket close to the DNA exit site. This domain shows flexibility with respect to the fixed zinc finger and moves with, but also relative to, the unwrapped DNA terminus. We apply molecular dynamics simulations of the histone tails in the nucleosome to show that in this mode of interaction, the active site of SIRT6 is perfectly poised to catalyze deacetylation of the H3 histone tail and that the partial unwrapping of the DNA allows even lysines close to the H3 core to reach the enzyme. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_18497.map.gz emd_18497.map.gz | 31.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-18497-v30.xml emd-18497-v30.xml emd-18497.xml emd-18497.xml | 15.4 KB 15.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_18497.png emd_18497.png | 64.6 KB | ||
| Filedesc metadata |  emd-18497.cif.gz emd-18497.cif.gz | 4.9 KB | ||
| Others |  emd_18497_half_map_1.map.gz emd_18497_half_map_1.map.gz emd_18497_half_map_2.map.gz emd_18497_half_map_2.map.gz | 59.5 MB 59.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18497 http://ftp.pdbj.org/pub/emdb/structures/EMD-18497 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18497 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18497 | HTTPS FTP |
-Validation report
| Summary document |  emd_18497_validation.pdf.gz emd_18497_validation.pdf.gz | 907.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_18497_full_validation.pdf.gz emd_18497_full_validation.pdf.gz | 907 KB | Display | |
| Data in XML |  emd_18497_validation.xml.gz emd_18497_validation.xml.gz | 12.3 KB | Display | |
| Data in CIF |  emd_18497_validation.cif.gz emd_18497_validation.cif.gz | 14.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18497 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18497 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18497 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18497 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_18497.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_18497.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.862 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_18497_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
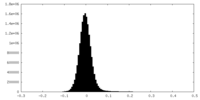
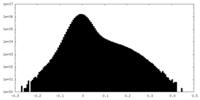
| Density Histograms |
-Half map: #2
| File | emd_18497_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
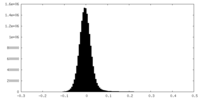
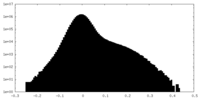
| Density Histograms |
- Sample components
Sample components
-Entire : Human Sirtuin 6 in complex with nucleosome
| Entire | Name: Human Sirtuin 6 in complex with nucleosome |
|---|---|
| Components |
|
-Supramolecule #1: Human Sirtuin 6 in complex with nucleosome
| Supramolecule | Name: Human Sirtuin 6 in complex with nucleosome / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Sirtuin 6
| Macromolecule | Name: Sirtuin 6 / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MSVNYAAGLS PYADKGKCGL PEIFDPPEEL ERKVWELARL VWQSSSVVFH TGAGISTASG IPDFRGPHG VWTMEERGLA PKFDTTFESA RPTQTHMALV QLERVGLLRF LVSQNVDGLH V RSGFPRDK LAELHGNMFV EECAKCKTQY VRDTVVGTMG LKATGRLCTV ...String: MSVNYAAGLS PYADKGKCGL PEIFDPPEEL ERKVWELARL VWQSSSVVFH TGAGISTASG IPDFRGPHG VWTMEERGLA PKFDTTFESA RPTQTHMALV QLERVGLLRF LVSQNVDGLH V RSGFPRDK LAELHGNMFV EECAKCKTQY VRDTVVGTMG LKATGRLCTV AKARGLRACR GE LRDTILD WEDSLPDRDL ALADEASRNA DLSITLGTSL QIRPSGNLPL ATKRRGGRLV IVN LQPTKH DRHADLRIHG YVDEVMTRLM KHLGLEIPAW DGPRVLERAL PPLPRPPTPK LEPK EESPT RINGSIPAGP KQEPCAQHNG SEPASPKRER PTSPAPHRPP KRVKAKAVPS |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Details: 20 mM Hepes pH 7.5, 2% glycerol v/v, 0.5 mM TCEP and 150 mM NaCl |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 2 / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 90 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 279 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 48.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.2 µm / Nominal defocus min: 1.1 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER / Details: Ab-initio in cryoSPARC |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.78 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 97164 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)