+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of CUL9-RBX1 ubiquitin E3 ligase complex in unneddylated and neddylated conformation - focused cullin dimer | |||||||||
 マップデータ マップデータ | DeepEMhancer map | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Cullin-RING RBR E3 Ligase / LIGASE | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報negative regulation of beige fat cell differentiation / cellular response to camptothecin / cullin-RING-type E3 NEDD8 transferase / NEDD8 transferase activity / cullin-RING ubiquitin ligase complex / Cul7-RING ubiquitin ligase complex / cellular response to chemical stress / Loss of Function of FBXW7 in Cancer and NOTCH1 Signaling / positive regulation of protein autoubiquitination / RNA polymerase II transcription initiation surveillance ...negative regulation of beige fat cell differentiation / cellular response to camptothecin / cullin-RING-type E3 NEDD8 transferase / NEDD8 transferase activity / cullin-RING ubiquitin ligase complex / Cul7-RING ubiquitin ligase complex / cellular response to chemical stress / Loss of Function of FBXW7 in Cancer and NOTCH1 Signaling / positive regulation of protein autoubiquitination / RNA polymerase II transcription initiation surveillance / protein neddylation / NEDD8 ligase activity / protein K27-linked ubiquitination / negative regulation of response to oxidative stress / VCB complex / Cul5-RING ubiquitin ligase complex / ubiquitin-ubiquitin ligase activity / ubiquitin-dependent protein catabolic process via the C-end degron rule pathway / regulation of mitotic nuclear division / SCF ubiquitin ligase complex / Cul2-RING ubiquitin ligase complex / Cul3-RING ubiquitin ligase complex / negative regulation of type I interferon production / SCF-dependent proteasomal ubiquitin-dependent protein catabolic process / Cul4A-RING E3 ubiquitin ligase complex / Cul4-RING E3 ubiquitin ligase complex / Prolactin receptor signaling / negative regulation of mitophagy / Cul4B-RING E3 ubiquitin ligase complex / TGF-beta receptor signaling activates SMADs / regulation of proteolysis / regulation of postsynapse assembly / cullin family protein binding / anatomical structure morphogenesis / protein monoubiquitination / site of DNA damage / signal transduction in response to DNA damage / Nuclear events stimulated by ALK signaling in cancer / protein K48-linked ubiquitination / transcription-coupled nucleotide-excision repair / negative regulation of insulin receptor signaling pathway / regulation of cellular response to insulin stimulus / positive regulation of TORC1 signaling / post-translational protein modification / T cell activation / protein modification process / Regulation of BACH1 activity / negative regulation of canonical NF-kappaB signal transduction / cellular response to amino acid stimulus / Degradation of DVL / Degradation of CRY and PER proteins / negative regulation of canonical Wnt signaling pathway / Iron uptake and transport / Degradation of GLI1 by the proteasome / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / Recognition of DNA damage by PCNA-containing replication complex / Negative regulation of NOTCH4 signaling / RING-type E3 ubiquitin transferase / Hedgehog 'on' state / Vif-mediated degradation of APOBEC3G / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / Ubiquitin-Mediated Degradation of Phosphorylated Cdc25A / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / NOTCH1 Intracellular Domain Regulates Transcription / Degradation of beta-catenin by the destruction complex / Evasion by RSV of host interferon responses / modification-dependent protein catabolic process / DNA Damage Recognition in GG-NER / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / microtubule cytoskeleton organization / Constitutive Signaling by NOTCH1 PEST Domain Mutants / Constitutive Signaling by NOTCH1 HD+PEST Domain Mutants / Dual Incision in GG-NER / Transcription-Coupled Nucleotide Excision Repair (TC-NER) / Formation of TC-NER Pre-Incision Complex / Regulation of expression of SLITs and ROBOs / Formation of Incision Complex in GG-NER / Interleukin-1 signaling / protein tag activity / Orc1 removal from chromatin / protein polyubiquitination / Dual incision in TC-NER / Regulation of RAS by GAPs / Gap-filling DNA repair synthesis and ligation in TC-NER / ubiquitin-protein transferase activity / Regulation of RUNX2 expression and activity / positive regulation of protein catabolic process / cellular response to UV / intracellular protein localization / ubiquitin protein ligase activity / UCH proteinases / KEAP1-NFE2L2 pathway / MAPK cascade / Cargo recognition for clathrin-mediated endocytosis / Antigen processing: Ubiquitination & Proteasome degradation / positive regulation of proteasomal ubiquitin-dependent protein catabolic process / Neddylation / cellular response to oxidative stress / spermatogenesis 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
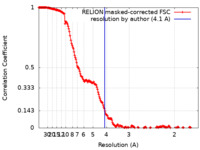
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.1 Å | |||||||||
 データ登録者 データ登録者 | Hopf LVM / Horn-Ghetko D / Schulman BA | |||||||||
| 資金援助 | European Union,  ドイツ, 2件 ドイツ, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2024 ジャーナル: Nat Struct Mol Biol / 年: 2024タイトル: Noncanonical assembly, neddylation and chimeric cullin-RING/RBR ubiquitylation by the 1.8 MDa CUL9 E3 ligase complex. 著者: Daniel Horn-Ghetko / Linus V M Hopf / Ishita Tripathi-Giesgen / Jiale Du / Sebastian Kostrhon / D Tung Vu / Viola Beier / Barbara Steigenberger / J Rajan Prabu / Luca Stier / Elias M Bruss / ...著者: Daniel Horn-Ghetko / Linus V M Hopf / Ishita Tripathi-Giesgen / Jiale Du / Sebastian Kostrhon / D Tung Vu / Viola Beier / Barbara Steigenberger / J Rajan Prabu / Luca Stier / Elias M Bruss / Matthias Mann / Yue Xiong / Brenda A Schulman /   要旨: Ubiquitin ligation is typically executed by hallmark E3 catalytic domains. Two such domains, 'cullin-RING' and 'RBR', are individually found in several hundred human E3 ligases, and collaborate with ...Ubiquitin ligation is typically executed by hallmark E3 catalytic domains. Two such domains, 'cullin-RING' and 'RBR', are individually found in several hundred human E3 ligases, and collaborate with E2 enzymes to catalyze ubiquitylation. However, the vertebrate-specific CUL9 complex with RBX1 (also called ROC1), of interest due to its tumor suppressive interaction with TP53, uniquely encompasses both cullin-RING and RBR domains. Here, cryo-EM, biochemistry and cellular assays elucidate a 1.8-MDa hexameric human CUL9-RBX1 assembly. Within one dimeric subcomplex, an E2-bound RBR domain is activated by neddylation of its own cullin domain and positioning from the adjacent CUL9-RBX1 in trans. Our data show CUL9 as unique among RBX1-bound cullins in dependence on the metazoan-specific UBE2F neddylation enzyme, while the RBR domain protects it from deneddylation. Substrates are recruited to various upstream domains, while ubiquitylation relies on both CUL9's neddylated cullin and RBR domains achieving self-assembled and chimeric cullin-RING/RBR E3 ligase activity. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
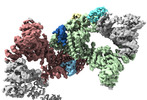
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_18216.map.gz emd_18216.map.gz | 433.7 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-18216-v30.xml emd-18216-v30.xml emd-18216.xml emd-18216.xml | 24.6 KB 24.6 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_18216_fsc.xml emd_18216_fsc.xml | 17.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_18216.png emd_18216.png | 133.5 KB | ||
| マスクデータ |  emd_18216_msk_1.map emd_18216_msk_1.map | 488.4 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-18216.cif.gz emd-18216.cif.gz | 7.8 KB | ||
| その他 |  emd_18216_additional_1.map.gz emd_18216_additional_1.map.gz emd_18216_half_map_1.map.gz emd_18216_half_map_1.map.gz emd_18216_half_map_2.map.gz emd_18216_half_map_2.map.gz | 31 MB 391.2 MB 390.4 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18216 http://ftp.pdbj.org/pub/emdb/structures/EMD-18216 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18216 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18216 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8q7hMC  8q7eC  8rhzC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_18216.map.gz / 形式: CCP4 / 大きさ: 488.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_18216.map.gz / 形式: CCP4 / 大きさ: 488.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | DeepEMhancer map | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.8512 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
| ファイル |  emd_18216_msk_1.map emd_18216_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: postprocess map
| ファイル | emd_18216_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | postprocess map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_18216_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_18216_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Structure of CUL9-RBX1 ubiquitin E3 ligase complex in unneddylate...
| 全体 | 名称: Structure of CUL9-RBX1 ubiquitin E3 ligase complex in unneddylated and neddylated conformation - focused cullin dimer |
|---|---|
| 要素 |
|
-超分子 #1: Structure of CUL9-RBX1 ubiquitin E3 ligase complex in unneddylate...
| 超分子 | 名称: Structure of CUL9-RBX1 ubiquitin E3 ligase complex in unneddylated and neddylated conformation - focused cullin dimer タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#4 / 詳細: Structure of dimeric subunit |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Cullin-9
| 分子 | 名称: Cullin-9 / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 281.686062 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: GPMVGERHAG DLMVPLGPRL QAYPEELIRQ RPGHDGHPEY LIRWSVLKCG EVGKVGVEEG KAEHILMWLS APEVYANCPG LLGERALSK GLQHEPAGVS GSFPRDPGGL DEVAMGEMEA DVQALVRRAA RQLAESGTPS LTAAVLHTIH VLSAYASIGP L TGVFRETG ...文字列: GPMVGERHAG DLMVPLGPRL QAYPEELIRQ RPGHDGHPEY LIRWSVLKCG EVGKVGVEEG KAEHILMWLS APEVYANCPG LLGERALSK GLQHEPAGVS GSFPRDPGGL DEVAMGEMEA DVQALVRRAA RQLAESGTPS LTAAVLHTIH VLSAYASIGP L TGVFRETG ALDLLMHMLC NPEPQIRRSA GKMLQALAAH DAGSRAHVLL SLSQQDGIEQ HMDFDSRYTL LELFAETTSS EE HCMAFEG IHLPQIPGKL LFSLVKRYLC VTSLLDQLNS SPELGAGDQS SPCATREKSR GQRELEFSMA VGNLISELVR SMG WARNLS EQGMSPPRPT RSIFQPYISG PSLLLPTIVT TPRRQGWVFR QRSEFSSRSG YGEYVQQTLQ PGMRVRMLDD YEEI SAGDE GEFRQSNNGI PPVQVFWQST GRTYWVHWHM LEILGPEEAT EDKASAAVEK GAGATVLGTA FPSWDWNPMD GLYPL PYLQ PEPQKNERVG YLTQAEWWEL LFFIKKLDLC EQQPIFQNLW KNLDETLGEK ALGEISVSVE MAESLLQVLS SRFEGS TLN DLLNSQIYTK YGLLSNEPSS SSTSRNHSCT PDPEEESKSE ASFSEEETES LKAKAEAPKT EAEPTKTRTE TPMAQSD SQ LFNQLLVTEG MTLPTEMKEA ASEMARALRG PGPRSSLDQH VAAVVATVQI SSLDTNLQLS GLSALSQAVE EVTERDHP L VRPDRSLREK LVKMLVELLT NQVGEKMVVV QALRLLYLLM TKHEWRPLFA REGGIYAVLV CMQEYKTSVL VQQAGLAAL KMLAVASSSE IPTFVTGRDS IHSLFDAQMT REIFASIDSA TRPGSESLLL TVPAAVILML NTEGCSSAAR NGLLLLNLLL CNHHTLGDQ IITQELRDTL FRHSGIAPRT EPMPTTRTIL MMLLNRYSEP PGSPERAALE TPIIQGQDGS PELLIRSLVG G PSAELLLD LERVLCREGS PGGAVRPLLK RLQQETQPFL LLLRTLDAPG PNKTLLLSVL RVITRLLDFP EAMVLPWHEV LE PCLNCLS GPSSDSEIVQ ELTCFLHRLA SMHKDYAVVL CCLGAKEILS KVLDKHSAQL LLGCELRDLV TECEKYAQLY SNL TSSILA GCIQMVLGQI EDHRRTHQPI NIPFFDVFLR HLCQGSSVEV KEDKCWEKVE VSSNPHRASK LTDHNPKTYW ESNG STGSH YITLHMHRGV LVRQLTLLVA SEDSSYMPAR VVVFGGDSTS CIGTELNTVN VMPSASRVIL LENLNRFWPI IQIRI KRCQ QGGIDTRVRG VEVLGPKPTF WPLFREQLCR RTCLFYTIRA QAWSRDIAED HRRLLQLCPR LNRVLRHEQN FADRFL PDD EAAQALGKTC WEALVSPLVQ NITSPDAEGV SALGWLLDQY LEQRETSRNP LSRAASFASR VRRLCHLLVH VEPPPGP SP EPSTRPFSKN SKGRDRSPAP SPVLPSSSLR NITQCWLSVV QEQVSRFLAA AWRAPDFVPR YCKLYEHLQR AGSELFGP R AAFMLALRSG FSGALLQQSF LTAAHMSEQF ARYIDQQIQG GLIGGAPGVE MLGQLQRHLE PIMVLSGLEL ATTFEHFYQ HYMADRLLSF GSSWLEGAVL EQIGLCFPNR LPQLMLQSLS TSEELQRQFH LFQLQRLDKL FLEQEDEEEK RLEEEEEEEE EEEAEKELF IEDPSPAISI LVLSPRCWPV SPLCYLYHPR KCLPTEFCDA LDRFSSFYSQ SQNHPVLDMG PHRRLQWTWL G RAELQFGK QILHVSTVQM WLLLKFNQTE EVSVETLLKD SDLSPELLLQ ALVPLTSGNG PLTLHEGQDF PHGGVLRLHE PG PQRSGEA LWLIPPQAYL NVEKDEGRTL EQKRNLLSCL LVRILKAHGE KGLHIDQLVC LVLEAWQKGP NPPGTLGHTV AGG VACTST DVLSCILHLL GQGYVKRRDD RPQILMYAAP EPMGPCRGQA DVPFCGSQSE TSKPSPEAVA TLASLQLPAG RTMS PQEVE GLMKQTVRQV QETLNLEPDV AQHLLAHSHW GAEQLLQSYS EDPEPLLLAA GLCVHQAQAV PVRPDHCPVC VSPLG CDDD LPSLCCMHYC CKSCWNEYLT TRIEQNLVLN CTCPIADCPA QPTGAFIRAI VSSPEVISKY EKALLRGYVE SCSNLT WCT NPQGCDRILC RQGLGCGTTC SKCGWASCFN CSFPEAHYPA SCGHMSQWVD DGGYYDGMSV EAQSKHLAKL ISKRCPS CQ APIEKNEGCL HMTCAKCNHG FCWRCLKSWK PNHKDYYNCS AMVSKAARQE KRFQDYNERC TFHHQAREFA VNLRNRVS A IHEVPPPRSF TFLNDACQGL EQARKVLAYA CVYSFYSQDA EYMDVVEQQT ENLELHTNAL QILLEETLLR CRDLASSLR LLRADCLSTG MELLRRIQER LLAILQHSAQ DFRVGLQSPS VEAWEAKGPN MPGSQPQASS GPEAEEEEED DEDDVPEWQQ DEFDEELDN DSFSYDESEN LDQETFFFGD EEEDEDEAYD UniProtKB: Cullin-9 |
-分子 #2: E3 ubiquitin-protein ligase RBX1
| 分子 | 名称: E3 ubiquitin-protein ligase RBX1 / タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO / EC番号: RING-type E3 ubiquitin transferase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 12.289977 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MAAAMDVDTP SGTNSGAGKK RFEVKKWNAV ALWAWDIVVD NCAICRNHIM DLCIECQANQ ASATSEECTV AWGVCNHAFH FHCISRWLK TRQVCPLDNR EWEFQKYGH UniProtKB: E3 ubiquitin-protein ligase RBX1 |
-分子 #3: NEDD8
| 分子 | 名称: NEDD8 / タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 9.086562 KDa |
| 配列 | 文字列: MLIKVKTLTG KEIEIDIEPT DKVERIKERV EEKEGIPPQQ QRLIYSGKQM NDEKTAADYK ILGGSVLHLV LALRGGGGLR Q UniProtKB: Ubiquitin-like protein NEDD8 |
-分子 #4: Unknown
| 分子 | 名称: Unknown / タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 12.868854 KDa |
| 配列 | 文字列: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) ...文字列: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) |
-分子 #5: ZINC ION
| 分子 | 名称: ZINC ION / タイプ: ligand / ID: 5 / コピー数: 10 / 式: ZN |
|---|---|
| 分子量 | 理論値: 65.409 Da |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 3 mg/mL |
|---|---|
| 緩衝液 | pH: 7.5 |
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) 平均電子線量: 60.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD 最大 デフォーカス(公称値): 2.8000000000000003 µm 最小 デフォーカス(公称値): 0.7000000000000001 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)