+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1746 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | P22 F170L C1 polyheads | |||||||||
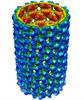
 Map data Map data | This is a helical reconstruction of a P22 polyhead with F170L coat protein with C1 symmetry | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | cryo-TEM / image reconstruction / helical reconstruction / bacteriophage P22 / virus assembly / coat protein | |||||||||
| Biological species |  Enterobacteria phage P22 (virus) Enterobacteria phage P22 (virus) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 13.0 Å | |||||||||
 Authors Authors | Parent KN / Sinkovits RS / Suhanovsky MM / Teschke CM / Egelman EH / Baker TS | |||||||||
 Citation Citation |  Journal: Phys Biol / Year: 2010 Journal: Phys Biol / Year: 2010Title: Cryo-reconstructions of P22 polyheads suggest that phage assembly is nucleated by trimeric interactions among coat proteins. Authors: Kristin N Parent / Robert S Sinkovits / Margaret M Suhanovsky / Carolyn M Teschke / Edward H Egelman / Timothy S Baker /  Abstract: Bacteriophage P22 forms an isometric capsid during normal assembly, yet when the coat protein (CP) is altered at a single site, helical structures (polyheads) also form. The structures of three ...Bacteriophage P22 forms an isometric capsid during normal assembly, yet when the coat protein (CP) is altered at a single site, helical structures (polyheads) also form. The structures of three distinct polyheads obtained from F170L and F170A variants were determined by cryo-reconstruction methods. An understanding of the structures of aberrant assemblies such as polyheads helps to explain how amino acid substitutions affect the CP, and these results can now be put into the context of CP pseudo-atomic models. F170L CP forms two types of polyhead and each has the CP organized as hexons (oligomers of six CPs). These hexons have a skewed structure similar to that in procapsids (precursor capsids formed prior to dsDNA packaging), yet their organization differs completely in polyheads and procapsids. F170A CP forms only one type of polyhead, and though this has hexons organized similarly to hexons in F170L polyheads, the hexons are isometric structures like those found in mature virions. The hexon organization in all three polyheads suggests that nucleation of procapsid assembly occurs via a trimer of CP monomers, and this drives formation of a T = 7, isometric particle. These variants also form procapsids, but they mature quite differently: F170A expands spontaneously at room temperature, whereas F170L requires more energy. The P22 CP structure along with scaffolding protein interactions appear to dictate curvature and geometry in assembled structures and residue 170 significantly influences both assembly and maturation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1746.map.gz emd_1746.map.gz | 167.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1746-v30.xml emd-1746-v30.xml emd-1746.xml emd-1746.xml | 10.3 KB 10.3 KB | Display Display |  EMDB header EMDB header |
| Images |  1746.png 1746.png | 570.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1746 http://ftp.pdbj.org/pub/emdb/structures/EMD-1746 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1746 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1746 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_1746.map.gz / Format: CCP4 / Size: 173.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1746.map.gz / Format: CCP4 / Size: 173.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is a helical reconstruction of a P22 polyhead with F170L coat protein with C1 symmetry | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.883 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : F170L coat protein in C1 helical lattice
| Entire | Name: F170L coat protein in C1 helical lattice |
|---|---|
| Components |
|
-Supramolecule #1000: F170L coat protein in C1 helical lattice
| Supramolecule | Name: F170L coat protein in C1 helical lattice / type: sample / ID: 1000 Details: This sample was generated by concentrating coat protein monomers. The asymmetric unit is a hexon. Number unique components: 1 |
|---|
-Supramolecule #1: Enterobacteria phage P22
| Supramolecule | Name: Enterobacteria phage P22 / type: virus / ID: 1 / Name.synonym: Coat protein / NCBI-ID: 10754 / Sci species name: Enterobacteria phage P22 / Database: NCBI / Virus type: OTHER / Virus isolate: STRAIN / Virus enveloped: No / Virus empty: Yes / Syn species name: Coat protein |
|---|---|
| Host (natural) | Organism:  Salmonella enterica subsp. enterica serovar Typhimurium (bacteria) Salmonella enterica subsp. enterica serovar Typhimurium (bacteria)synonym: BACTERIA(EUBACTERIA) |
| Molecular weight | Experimental: 47 MDa / Theoretical: 47 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 35 mg/mL |
|---|---|
| Buffer | pH: 7.6 / Details: 20 mM sodium phosphate |
| Grid | Details: Lacey-carbon |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 89 K / Instrument: HOMEMADE PLUNGER / Details: Vitrification instrument: Manual plunge-freezer / Method: Blot for 5 seconds before freezing |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Temperature | Min: 90 K / Max: 90 K / Average: 90 K |
| Alignment procedure | Legacy - Astigmatism: At working magnification |
| Date | Oct 10, 2008 |
| Image recording | Category: CCD / Film or detector model: GENERIC GATAN (4k x 4k) / Digitization - Sampling interval: 1.883 µm / Number real images: 1141 / Average electron dose: 19 e/Å2 / Od range: 1.5 / Bits/pixel: 16 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 79666 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.3 mm / Nominal defocus max: 3.39 µm / Nominal defocus min: 1.14 µm / Nominal magnification: 79666 |
| Sample stage | Specimen holder: Polara Multi Specimen Holder / Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | This reconstruction was done with the iterative helical real space reconstruction method |
|---|---|
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 10.5 Å Applied symmetry - Helical parameters - Δ&Phi: 37.4 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 13.0 Å / Resolution method: OTHER / Software - Name: IHRSR |
| CTF correction | Details: ROBEM |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















