[English] 日本語
 Yorodumi
Yorodumi- EMDB-15950: Direct observation of the closed conformational state of formin m... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Direct observation of the closed conformational state of formin mDia1 at actin filament barbed end | |||||||||
 Map data Map data | Actin barbed-end - Formin mDia1 "closed state" | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Actin binding protein / Complex / barbed-end / formin / MOTOR PROTEIN | |||||||||
| Biological species |   | |||||||||
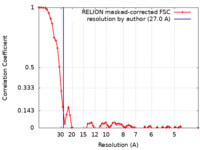
| Method | single particle reconstruction / negative staining / Resolution: 27.0 Å | |||||||||
 Authors Authors | Maufront J / Guichard B / Cao L / Di Cicco A / Jegou A / Romet-Lemonne G / Bertin A | |||||||||
| Funding support |  France, 1 items France, 1 items
| |||||||||
 Citation Citation |  Journal: Mol Biol Cell / Year: 2023 Journal: Mol Biol Cell / Year: 2023Title: Direct observation of the conformational states of formin mDia1 at actin filament barbed ends and along the filament. Authors: Julien Maufront / Bérengère Guichard / Lu-Yan Cao / Aurélie Di Cicco / Antoine Jégou / Guillaume Romet-Lemonne / Aurélie Bertin /  Abstract: The fine regulation of actin polymerization is essential to control cell motility and architecture and to perform essential cellular functions. Formins are key regulators of actin filament assembly, ...The fine regulation of actin polymerization is essential to control cell motility and architecture and to perform essential cellular functions. Formins are key regulators of actin filament assembly, known to processively elongate filament barbed ends and increase their polymerization rate. Different models have been extrapolated to describe the molecular mechanism governing the processive motion of formin FH2 domains at polymerizing barbed ends. Using negative stain electron microscopy, we directly identified for the first time two conformations of the mDia1 formin FH2 domains in interaction with the barbed ends of actin filaments. These conformations agree with the speculated open and closed conformations of the "stair-stepping" model. We observed the FH2 dimers to be in the open conformation for 79% of the data, interacting with the two terminal actin subunits of the barbed end while they interact with three actin subunits in the closed conformation. In addition, we identified and characterized the structure of single FH2 dimers encircling the core of actin filaments, and reveal their ability to spontaneously depart from barbed ends. #1:  Journal: BiorXiv / Year: 2022 Journal: BiorXiv / Year: 2022Title: Direct observation of the open conformational state of formin mDia1 at actin filament barbed end Authors: Maufront J / Bertin A | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15950.map.gz emd_15950.map.gz | 1.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15950-v30.xml emd-15950-v30.xml emd-15950.xml emd-15950.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_15950_fsc.xml emd_15950_fsc.xml | 6.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_15950.png emd_15950.png | 32.2 KB | ||
| Masks |  emd_15950_msk_1.map emd_15950_msk_1.map | 22.2 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-15950.cif.gz emd-15950.cif.gz | 4.4 KB | ||
| Others |  emd_15950_half_map_1.map.gz emd_15950_half_map_1.map.gz emd_15950_half_map_2.map.gz emd_15950_half_map_2.map.gz | 17 MB 17 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15950 http://ftp.pdbj.org/pub/emdb/structures/EMD-15950 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15950 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15950 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15950.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15950.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Actin barbed-end - Formin mDia1 "closed state" | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.13 Å | ||||||||||||||||||||||||||||||||||||
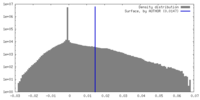
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_15950_msk_1.map emd_15950_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
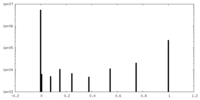
| Density Histograms |
-Half map: Actin barbed-end - Formin mDia1 "closed state"
| File | emd_15950_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Actin barbed-end - Formin mDia1 "closed state" | ||||||||||||
| Projections & Slices |
| ||||||||||||
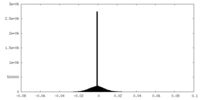
| Density Histograms |
-Half map: Actin barbed-end - Formin mDia1 "closed state"
| File | emd_15950_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Actin barbed-end - Formin mDia1 "closed state" | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Actin barbed-end bound by formin mDia1 dimer in his "closed state"
| Entire | Name: Actin barbed-end bound by formin mDia1 dimer in his "closed state" |
|---|---|
| Components |
|
-Supramolecule #1: Actin barbed-end bound by formin mDia1 dimer in his "closed state"
| Supramolecule | Name: Actin barbed-end bound by formin mDia1 dimer in his "closed state" type: complex / ID: 1 / Parent: 0 |
|---|
-Supramolecule #2: Formin mDia1 dimer
| Supramolecule | Name: Formin mDia1 dimer / type: organelle_or_cellular_component / ID: 2 / Parent: 1 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: Alpha Actin subunits assembled in helicoidal F-actin filament
| Supramolecule | Name: Alpha Actin subunits assembled in helicoidal F-actin filament type: organelle_or_cellular_component / ID: 3 / Parent: 1 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation #1
Sample preparation #1
| Preparation ID | 1 |
|---|---|
| Concentration | 0.04 mg/mL |
| Buffer | pH: 7.8 |
| Staining | Type: NEGATIVE / Material: Uranyl Formate |
- Sample preparation #2
Sample preparation #2
| Preparation ID | 2 |
|---|---|
| Concentration | 0.01 mg/mL |
| Buffer | pH: 7.8 |
| Staining | Type: NEGATIVE / Material: Uranyl Formate |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 20 |
|---|---|
| Image recording | Film or detector model: TVIPS TEMCAM-F416 (4k x 4k) / Average electron dose: 20.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source: LAB6 |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)