[English] 日本語
 Yorodumi
Yorodumi- EMDB-15853: Tetrameric structure of 47 N-terminally truncated human tryptopha... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Tetrameric structure of 47 N-terminally truncated human tryptophan hydroxylase 2 with dimerized regulatory domains | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Tryptophan hydroxylase 2 / serotonin biosynthesis / OXIDOREDUCTASE | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 8.5 Å | |||||||||
 Authors Authors | Zhang Z / Vedel IM / Skawinska NT / Harris P / Stark H / Peters GHJ | |||||||||
| Funding support |  Denmark, 1 items Denmark, 1 items
| |||||||||
 Citation Citation |  Journal: Structure / Year: 2023 Journal: Structure / Year: 2023Title: Structural characterization of human tryptophan hydroxylase 2 reveals that L-Phe is superior to L-Trp as the regulatory domain ligand. Authors: Ida M Vedel / Andreas Prestel / Zhenwei Zhang / Natalia T Skawinska / Holger Stark / Pernille Harris / Birthe B Kragelund / Günther H J Peters /   Abstract: Tryptophan hydroxylase 2 (TPH2) catalyzes the rate-limiting step in serotonin biosynthesis in the brain. Consequently, regulation of TPH2 is relevant for serotonin-related diseases, yet the ...Tryptophan hydroxylase 2 (TPH2) catalyzes the rate-limiting step in serotonin biosynthesis in the brain. Consequently, regulation of TPH2 is relevant for serotonin-related diseases, yet the regulatory mechanism of TPH2 is poorly understood and structural and dynamical insights are missing. We use NMR spectroscopy to determine the structure of a 47 N-terminally truncated variant of the regulatory domain (RD) dimer of human TPH2 in complex with L-Phe, and show that L-Phe is the superior RD ligand compared with the natural substrate, L-Trp. Using cryo-EM, we obtain a low-resolution structure of a similarly truncated variant of the complete tetrameric enzyme with dimerized RDs. The cryo-EM two-dimensional (2D) class averages additionally indicate that the RDs are dynamic in the tetramer and likely exist in a monomer-dimer equilibrium. Our results provide structural information on the RD as an isolated domain and in the TPH2 tetramer, which will facilitate future elucidation of TPH2's regulatory mechanism. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15853.map.gz emd_15853.map.gz | 1.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15853-v30.xml emd-15853-v30.xml emd-15853.xml emd-15853.xml | 12.4 KB 12.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15853.png emd_15853.png | 45.8 KB | ||
| Others |  emd_15853_half_map_1.map.gz emd_15853_half_map_1.map.gz emd_15853_half_map_2.map.gz emd_15853_half_map_2.map.gz | 1.4 MB 1.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15853 http://ftp.pdbj.org/pub/emdb/structures/EMD-15853 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15853 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15853 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15853.map.gz / Format: CCP4 / Size: 2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15853.map.gz / Format: CCP4 / Size: 2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.12 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_15853_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
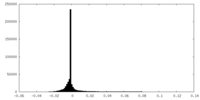
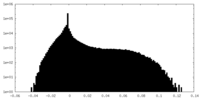
| Density Histograms |
-Half map: #2
| File | emd_15853_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
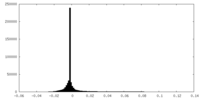
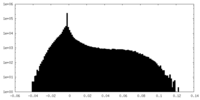
| Density Histograms |
- Sample components
Sample components
-Entire : Tryptophan 5-hydroxylase 2
| Entire | Name: Tryptophan 5-hydroxylase 2 |
|---|---|
| Components |
|
-Supramolecule #1: Tryptophan 5-hydroxylase 2
| Supramolecule | Name: Tryptophan 5-hydroxylase 2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: INTEGRATING / Average electron dose: 78.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 4.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER / Details: ab initio 3D |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: FOURIER SPACE / Resolution.type: BY AUTHOR / Resolution: 8.5 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 58616 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)