+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of ex vivo AA amyloid from renal tissue of a short hair cat deceased in a shelter | |||||||||
 マップデータ マップデータ | autosharpened map | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | AA amyloidosis / cat / Serum amyloid A / PROTEIN FIBRIL | |||||||||
| 機能・相同性 | Serum amyloid A protein / : / Serum amyloid A protein / Serum amyloid A proteins signature. / Serum amyloid A proteins / high-density lipoprotein particle / acute-phase response / Serum amyloid A protein 機能・相同性情報 機能・相同性情報 | |||||||||
| 生物種 |  | |||||||||
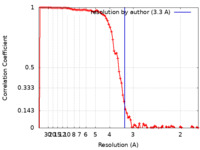
| 手法 | らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 3.3 Å | |||||||||
 データ登録者 データ登録者 | Schulte T / Chaves-Sanjuan A / Ricagno S | |||||||||
| 資金援助 |  イタリア, 1件 イタリア, 1件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2022 ジャーナル: Nat Commun / 年: 2022タイトル: Cryo-EM structure of ex vivo fibrils associated with extreme AA amyloidosis prevalence in a cat shelter. 著者: Tim Schulte / Antonio Chaves-Sanjuan / Giulia Mazzini / Valentina Speranzini / Francesca Lavatelli / Filippo Ferri / Carlo Palizzotto / Maria Mazza / Paolo Milani / Mario Nuvolone / Anne- ...著者: Tim Schulte / Antonio Chaves-Sanjuan / Giulia Mazzini / Valentina Speranzini / Francesca Lavatelli / Filippo Ferri / Carlo Palizzotto / Maria Mazza / Paolo Milani / Mario Nuvolone / Anne-Cathrine Vogt / Monique Vogel / Giovanni Palladini / Giampaolo Merlini / Martino Bolognesi / Silvia Ferro / Eric Zini / Stefano Ricagno /   要旨: AA amyloidosis is a systemic disease characterized by deposition of misfolded serum amyloid A protein (SAA) into cross-β amyloid in multiple organs in humans and animals. AA amyloidosis occurs at ...AA amyloidosis is a systemic disease characterized by deposition of misfolded serum amyloid A protein (SAA) into cross-β amyloid in multiple organs in humans and animals. AA amyloidosis occurs at high SAA serum levels during chronic inflammation. Prion-like transmission was reported as possible cause of extreme AA amyloidosis prevalence in captive animals, e.g. 70% in cheetah and 57-73% in domestic short hair (DSH) cats kept in zoos and shelters, respectively. Herein, we present the 3.3 Å cryo-EM structure of AA amyloid extracted post-mortem from the kidney of a DSH cat with renal failure, deceased in a shelter with extreme disease prevalence. The structure reveals a cross-β architecture assembled from two 76-residue long proto-filaments. Despite >70% sequence homology to mouse and human SAA, the cat SAA variant adopts a distinct amyloid fold. Inclusion of an eight-residue insert unique to feline SAA contributes to increased amyloid stability. The presented feline AA amyloid structure is fully compatible with the 99% identical amino acid sequence of amyloid fragments of captive cheetah. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_14726.map.gz emd_14726.map.gz | 53.8 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-14726-v30.xml emd-14726-v30.xml emd-14726.xml emd-14726.xml | 18.3 KB 18.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_14726_fsc.xml emd_14726_fsc.xml | 11.4 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_14726.png emd_14726.png | 135.6 KB | ||
| マスクデータ |  emd_14726_msk_1.map emd_14726_msk_1.map | 59.6 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-14726.cif.gz emd-14726.cif.gz | 5.6 KB | ||
| その他 |  emd_14726_additional_1.map.gz emd_14726_additional_1.map.gz emd_14726_half_map_1.map.gz emd_14726_half_map_1.map.gz emd_14726_half_map_2.map.gz emd_14726_half_map_2.map.gz | 44.1 MB 44.5 MB 44.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14726 http://ftp.pdbj.org/pub/emdb/structures/EMD-14726 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14726 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14726 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7zh7MC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_14726.map.gz / 形式: CCP4 / 大きさ: 59.6 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_14726.map.gz / 形式: CCP4 / 大きさ: 59.6 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | autosharpened map | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.889 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
| ファイル |  emd_14726_msk_1.map emd_14726_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
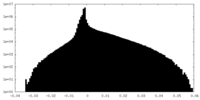
| 密度ヒストグラム |
-追加マップ: map after 3D autorefine with gold-standard solvent-flattened fsc
| ファイル | emd_14726_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | map after 3D autorefine with gold-standard solvent-flattened fsc | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
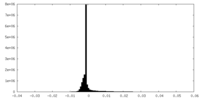
| 密度ヒストグラム |
-ハーフマップ: half map 2
| ファイル | emd_14726_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half map 2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
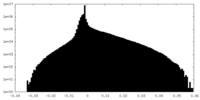
| 密度ヒストグラム |
-ハーフマップ: half map 1
| ファイル | emd_14726_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half map 1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : AA amyloid extracted from kidney of deceased cat
| 全体 | 名称: AA amyloid extracted from kidney of deceased cat |
|---|---|
| 要素 |
|
-超分子 #1: AA amyloid extracted from kidney of deceased cat
| 超分子 | 名称: AA amyloid extracted from kidney of deceased cat / タイプ: organelle_or_cellular_component / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: Serum amyloid A protein
| 分子 | 名称: Serum amyloid A protein / タイプ: protein_or_peptide / ID: 1 / コピー数: 10 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 14.449766 KDa |
| 配列 | 文字列: MKLFTGLVFC SLVLGVSSEW YSFLGEAAQG AWDMWRAYSD MREANYIGAD KYFHARGNYD AAQRGPGGAW AAKVISDARE NSQRVTDFF RHGNSGHGAE DSKADQEANE WGRSGKDPNH YRPEGLPDKY UniProtKB: Serum amyloid A protein |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | helical array |
- 試料調製
試料調製
| 緩衝液 | pH: 7 |
|---|---|
| グリッド | モデル: C-flat-1.2/1.3 / 材質: COPPER / メッシュ: 300 / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 時間: 30 sec. 詳細: glow discharged for 30s at 30mA using a GloQube system |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TALOS ARCTICA |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON III (4k x 4k) 検出モード: COUNTING / 撮影したグリッド数: 1 / 実像数: 2652 / 平均電子線量: 40.0 e/Å2 / 詳細: images takes for analysis |
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.2 µm / 最小 デフォーカス(公称値): 0.6 µm / 倍率(公称値): 120000 |
| 試料ステージ | ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Talos Arctica / 画像提供: FEI Company |
- 画像解析
画像解析
-原子モデル構築 1
| 精密化 | 空間: REAL / プロトコル: AB INITIO MODEL / 温度因子: 76.7 |
|---|---|
| 得られたモデル |  PDB-7zh7: |
 ムービー
ムービー コントローラー
コントローラー




 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)