[English] 日本語
 Yorodumi
Yorodumi- EMDB-13319: Human methemoglobin bound to Staphylococcus aureus hemophore IsdB -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human methemoglobin bound to Staphylococcus aureus hemophore IsdB | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Iron acquisition / Hemophore / Hemoglobin / NEAT domain / METAL TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationheme transmembrane transporter activity / Heme assimilation / cellular oxidant detoxification / nitric oxide transport / hemoglobin alpha binding / hemoglobin binding / haptoglobin-hemoglobin complex / renal absorption / hemoglobin complex / oxygen transport ...heme transmembrane transporter activity / Heme assimilation / cellular oxidant detoxification / nitric oxide transport / hemoglobin alpha binding / hemoglobin binding / haptoglobin-hemoglobin complex / renal absorption / hemoglobin complex / oxygen transport / Scavenging of heme from plasma / erythrocyte development / endocytic vesicle lumen / blood vessel diameter maintenance / oxygen carrier activity / hydrogen peroxide catabolic process / carbon dioxide transport / response to hydrogen peroxide / Heme signaling / Erythrocytes take up oxygen and release carbon dioxide / Erythrocytes take up carbon dioxide and release oxygen / Cytoprotection by HMOX1 / Late endosomal microautophagy / oxygen binding / regulation of blood pressure / platelet aggregation / Chaperone Mediated Autophagy / positive regulation of nitric oxide biosynthetic process / tertiary granule lumen / Factors involved in megakaryocyte development and platelet production / blood microparticle / ficolin-1-rich granule lumen / iron ion binding / inflammatory response / heme binding / Neutrophil degranulation / extracellular space / extracellular exosome / extracellular region / metal ion binding / membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  Staphylococcus aureus subsp. aureus MW2 (bacteria) Staphylococcus aureus subsp. aureus MW2 (bacteria) | |||||||||
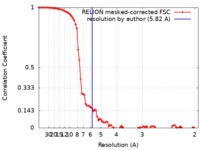
| Method | single particle reconstruction / cryo EM / Resolution: 5.82 Å | |||||||||
 Authors Authors | De Bei O / Chirgadze DY / Hardwick SW / Luisi BF / Campanini B | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2022 Journal: Proc Natl Acad Sci U S A / Year: 2022Title: Cryo-EM structures of staphylococcal IsdB bound to human hemoglobin reveal the process of heme extraction. Authors: Omar De Bei / Marialaura Marchetti / Luca Ronda / Eleonora Gianquinto / Loretta Lazzarato / Dimitri Y Chirgadze / Steven W Hardwick / Lee R Cooper / Francesca Spyrakis / Ben F Luisi / ...Authors: Omar De Bei / Marialaura Marchetti / Luca Ronda / Eleonora Gianquinto / Loretta Lazzarato / Dimitri Y Chirgadze / Steven W Hardwick / Lee R Cooper / Francesca Spyrakis / Ben F Luisi / Barbara Campanini / Stefano Bettati /   Abstract: Iron surface determinant B (IsdB) is a hemoglobin (Hb) receptor essential for hemic iron acquisition by Staphylococcus aureus. Heme transfer to IsdB is possible from oxidized Hb (metHb), but ...Iron surface determinant B (IsdB) is a hemoglobin (Hb) receptor essential for hemic iron acquisition by Staphylococcus aureus. Heme transfer to IsdB is possible from oxidized Hb (metHb), but inefficient from Hb either bound to oxygen (oxyHb) or bound to carbon monoxide (HbCO), and encompasses a sequence of structural events that are currently poorly understood. By single-particle cryo-electron microscopy, we determined the structure of two IsdB:Hb complexes, representing key species along the heme extraction pathway. The IsdB:HbCO structure, at 2.9-Å resolution, provides a snapshot of the preextraction complex. In this early stage of IsdB:Hb interaction, the hemophore binds to the β-subunits of the Hb tetramer, exploiting a folding-upon-binding mechanism that is likely triggered by a cis/trans isomerization of Pro173. Binding of IsdB to α-subunits occurs upon dissociation of the Hb tetramer into α/β dimers. The structure of the IsdB:metHb complex reveals the final step of the extraction process, where heme transfer to IsdB is completed. The stability of the complex, both before and after heme transfer from Hb to IsdB, is influenced by isomerization of Pro173. These results greatly enhance current understanding of structural and dynamic aspects of the heme extraction mechanism by IsdB and provide insight into the interactions that stabilize the complex before the heme transfer event. This information will support future efforts to identify inhibitors of heme acquisition by S. aureus by interfering with IsdB:Hb complex formation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13319.map.gz emd_13319.map.gz | 46 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13319-v30.xml emd-13319-v30.xml emd-13319.xml emd-13319.xml | 26.5 KB 26.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13319_fsc.xml emd_13319_fsc.xml | 9 KB | Display |  FSC data file FSC data file |
| Images |  emd_13319.png emd_13319.png | 108.9 KB | ||
| Masks |  emd_13319_msk_1.map emd_13319_msk_1.map | 59.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-13319.cif.gz emd-13319.cif.gz | 7.3 KB | ||
| Others |  emd_13319_additional_1.map.gz emd_13319_additional_1.map.gz emd_13319_additional_2.map.gz emd_13319_additional_2.map.gz emd_13319_half_map_1.map.gz emd_13319_half_map_1.map.gz emd_13319_half_map_2.map.gz emd_13319_half_map_2.map.gz | 30 MB 3.7 MB 46.2 MB 46.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13319 http://ftp.pdbj.org/pub/emdb/structures/EMD-13319 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13319 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13319 | HTTPS FTP |
-Related structure data
| Related structure data |  7pcfMC  7pchC  7pcqC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13319.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13319.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.978 Å | ||||||||||||||||||||||||||||||||||||
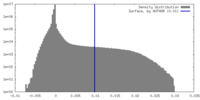
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_13319_msk_1.map emd_13319_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
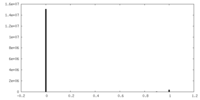
| Density Histograms |
-Additional map: This map was obtained using the local resolution tool in RELION-3.0.8
| File | emd_13319_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This map was obtained using the local resolution tool in RELION-3.0.8 | ||||||||||||
| Projections & Slices |
| ||||||||||||
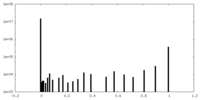
| Density Histograms |
-Additional map: This map is the result of CTF refinement,...
| File | emd_13319_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This map is the result of CTF refinement, Bayesian polishing, and post-processing in RELION-3.0.8 | ||||||||||||
| Projections & Slices |
| ||||||||||||
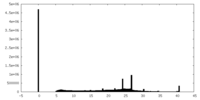
| Density Histograms |
-Half map: #2
| File | emd_13319_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_13319_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Human methemoglobin bound to Staphylococcus aureus hemophore IsdB
| Entire | Name: Human methemoglobin bound to Staphylococcus aureus hemophore IsdB |
|---|---|
| Components |
|
-Supramolecule #1: Human methemoglobin bound to Staphylococcus aureus hemophore IsdB
| Supramolecule | Name: Human methemoglobin bound to Staphylococcus aureus hemophore IsdB type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Molecular weight | Theoretical: 119 KDa |
-Supramolecule #2: Hemoglobin
| Supramolecule | Name: Hemoglobin / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: Iron-regulated surface determinant protein B
| Supramolecule | Name: Iron-regulated surface determinant protein B / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  Staphylococcus aureus subsp. aureus MW2 (bacteria) Staphylococcus aureus subsp. aureus MW2 (bacteria) |
-Macromolecule #1: Hemoglobin subunit alpha
| Macromolecule | Name: Hemoglobin subunit alpha / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 15.150353 KDa |
| Sequence | String: VLSPADKTNV KAAWGKVGAH AGEYGAEALE RMFLSFPTTK TYFPHFDLSH GSAQVKGHGK KVADALTNAV AHVDDMPNAL SALSDLHAH KLRVDPVNFK LLSHCLLVTL AAHLPAEFTP AVHASLDKFL ASVSTVLTSK YR UniProtKB: Hemoglobin subunit alpha |
-Macromolecule #2: Hemoglobin subunit beta
| Macromolecule | Name: Hemoglobin subunit beta / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 15.890198 KDa |
| Sequence | String: VHLTPEEKSA VTALWGKVNV DEVGGEALGR LLVVYPWTQR FFESFGDLST PDAVMGNPKV KAHGKKVLGA FSDGLAHLDN LKGTFATLS ELHCDKLHVD PENFRLLGNV LVCVLAHHFG KEFTPPVQAA YQKVVAGVAN ALAHKYH UniProtKB: Hemoglobin subunit beta |
-Macromolecule #3: Iron-regulated surface determinant protein B
| Macromolecule | Name: Iron-regulated surface determinant protein B / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Staphylococcus aureus subsp. aureus MW2 (bacteria) Staphylococcus aureus subsp. aureus MW2 (bacteria) |
| Molecular weight | Theoretical: 43.393129 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MLNQELREAI KNPAIKDKDH SAPNSRPIDF EMKKKDGTQQ FYHYASSVKP ARVIFTDSKP EIELGLQSGQ FWRKFEVYEG DKKLPIKLV SYDTVKDYAY IRFSVSNGTK AVKIVSSTHF NNKEEKYDYT LMEFAQPIYN SADKFKTEED YKAEKLLAPY K KAKTLERQ ...String: MLNQELREAI KNPAIKDKDH SAPNSRPIDF EMKKKDGTQQ FYHYASSVKP ARVIFTDSKP EIELGLQSGQ FWRKFEVYEG DKKLPIKLV SYDTVKDYAY IRFSVSNGTK AVKIVSSTHF NNKEEKYDYT LMEFAQPIYN SADKFKTEED YKAEKLLAPY K KAKTLERQ VYELNKIQDK LPEKLKAEYK KKLEDTKKAL DEQVKSAITE FQNVQPTNEK MTDLQDTKYV VYESVENNES MM DTFVKHP IKTGMLNGKK YMVMETTNDD YWKDFMVEGQ RVRTISKDAK NNTRTIIFPY VEGKTLYDAI VKVHVKTIDY DGQ YHVRIV DKEAFTKANT DKSNKKEQQD NSAKKEATPA TPSKPTSAWS HPQFEK UniProtKB: Iron-regulated surface determinant protein B |
-Macromolecule #4: PROTOPORPHYRIN IX CONTAINING FE
| Macromolecule | Name: PROTOPORPHYRIN IX CONTAINING FE / type: ligand / ID: 4 / Number of copies: 2 / Formula: HEM |
|---|---|
| Molecular weight | Theoretical: 616.487 Da |
| Chemical component information |  ChemComp-HEM: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.2 Component:
Details: CHAPSO was added immediately before plunge freezing to overcome preferred orientation of the particles in the vitreous ice. | |||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. / Pretreatment - Pressure: 38.5035 kPa / Details: The grid was discharged on both sides. | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 99 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 3051 / Average electron dose: 39.59 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.1 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 130000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)