+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-12963 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
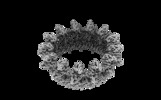
| タイトル | Structure of the outer-membrane core complex (outer ring) from a conjugative type IV secretion system | |||||||||
 マップデータ マップデータ | The structure of the outer-membrane core complex (outer ring) from a conjugative type IV secretion system | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Type IV secretion system / F plasmid / outer-membrane core complex / conjugation / MEMBRANE PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Pilus assembly TraK / Type-F conjugative transfer system secretin TraK / : / TraK N-terminal domain / TraK C-terminal domain / Type IV conjugative transfer system protein TraV / Type IV conjugative transfer system lipoprotein (TraV) / Prokaryotic membrane lipoprotein lipid attachment site profile. 類似検索 - ドメイン・相同性 | |||||||||
| 生物種 |  Salmonella enterica (サルモネラ菌) / Salmonella enterica (サルモネラ菌) /  Salmonella enterica subsp. salamae serovar 58:l,z13,z28:z6 (サルモネラ菌) Salmonella enterica subsp. salamae serovar 58:l,z13,z28:z6 (サルモネラ菌) | |||||||||
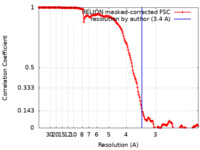
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.4 Å | |||||||||
 データ登録者 データ登録者 | Amin H / Ilangovan A / Costa TRD | |||||||||
| 資金援助 |  英国, 1件 英国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2021 ジャーナル: Nat Commun / 年: 2021タイトル: Architecture of the outer-membrane core complex from a conjugative type IV secretion system. 著者: Himani Amin / Aravindan Ilangovan / Tiago R D Costa /  要旨: Conjugation is one of the most important processes that bacteria utilize to spread antibiotic resistance genes among bacterial populations. Interbacterial DNA transfer requires a large double ...Conjugation is one of the most important processes that bacteria utilize to spread antibiotic resistance genes among bacterial populations. Interbacterial DNA transfer requires a large double membrane-spanning nanomachine called the type 4 secretion system (T4SS) made up of the inner-membrane complex (IMC), the outer-membrane core complex (OMCC) and the conjugative pilus. The iconic F plasmid-encoded T4SS has been central in understanding conjugation for several decades, however atomic details of its structure are not known. Here, we report the structure of a complete conjugative OMCC encoded by the pED208 plasmid from E. coli, solved by cryo-electron microscopy at 3.3 Å resolution. This 2.1 MDa complex has a unique arrangement with two radial concentric rings, each having a different symmetry eventually contributing to remarkable differences in protein stoichiometry and flexibility in comparison to other OMCCs. Our structure suggests that F-OMCC is a highly dynamic complex, with implications for pilus extension and retraction during conjugation. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_12963.map.gz emd_12963.map.gz | 391.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-12963-v30.xml emd-12963-v30.xml emd-12963.xml emd-12963.xml | 11 KB 11 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_12963_fsc.xml emd_12963_fsc.xml | 16.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_12963.png emd_12963.png | 63.9 KB | ||
| Filedesc metadata |  emd-12963.cif.gz emd-12963.cif.gz | 5.2 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12963 http://ftp.pdbj.org/pub/emdb/structures/EMD-12963 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12963 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12963 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_12963_validation.pdf.gz emd_12963_validation.pdf.gz | 663.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_12963_full_validation.pdf.gz emd_12963_full_validation.pdf.gz | 662.9 KB | 表示 | |
| XML形式データ |  emd_12963_validation.xml.gz emd_12963_validation.xml.gz | 15.6 KB | 表示 | |
| CIF形式データ |  emd_12963_validation.cif.gz emd_12963_validation.cif.gz | 21.3 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12963 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12963 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12963 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12963 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_12963.map.gz / 形式: CCP4 / 大きさ: 421.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_12963.map.gz / 形式: CCP4 / 大きさ: 421.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | The structure of the outer-membrane core complex (outer ring) from a conjugative type IV secretion system | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Outer-membrane core complex (outer ring)
| 全体 | 名称: Outer-membrane core complex (outer ring) |
|---|---|
| 要素 |
|
-超分子 #1: Outer-membrane core complex (outer ring)
| 超分子 | 名称: Outer-membrane core complex (outer ring) / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#2 |
|---|---|
| 由来(天然) | 生物種:  Salmonella enterica (サルモネラ菌) Salmonella enterica (サルモネラ菌) |
-分子 #1: Type IV conjugative transfer system lipoprotein TraV
| 分子 | 名称: Type IV conjugative transfer system lipoprotein TraV タイプ: protein_or_peptide / ID: 1 / コピー数: 26 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Salmonella enterica (サルモネラ菌) Salmonella enterica (サルモネラ菌) |
| 分子量 | 理論値: 20.92865 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MKKITLLLAG SALLLSGCAG VKSSFDCDAT TSDTCMTMTK ANQLARDKAA KQAGKPAAGG LPSLVNLPAT SAVEVPSASR SAVTPPSGT RTVSTTPPVS AGTSAGVNTN TTTSTLTPRP VAGTPVTTTP SSVAYRPVVS VVTPTPSCQN VRCDNPGTVH P QRSRDQIA ...文字列: MKKITLLLAG SALLLSGCAG VKSSFDCDAT TSDTCMTMTK ANQLARDKAA KQAGKPAAGG LPSLVNLPAT SAVEVPSASR SAVTPPSGT RTVSTTPPVS AGTSAGVNTN TTTSTLTPRP VAGTPVTTTP SSVAYRPVVS VVTPTPSCQN VRCDNPGTVH P QRSRDQIA TVWIAPWVDS DNAFHQPGRV SFVVSPADWV LPARVN UniProtKB: Type IV conjugative transfer system lipoprotein TraV |
-分子 #2: TraB
| 分子 | 名称: TraB / タイプ: protein_or_peptide / ID: 2 / コピー数: 13 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Salmonella enterica (サルモネラ菌) Salmonella enterica (サルモネラ菌) |
| 分子量 | 理論値: 1.128233 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: PGMMDSQEFS |
-分子 #3: Type-F conjugative transfer system secretin TraK
| 分子 | 名称: Type-F conjugative transfer system secretin TraK / タイプ: protein_or_peptide / ID: 3 / コピー数: 26 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Salmonella enterica subsp. salamae serovar 58:l,z13,z28:z6 (サルモネラ菌) Salmonella enterica subsp. salamae serovar 58:l,z13,z28:z6 (サルモネラ菌) |
| 分子量 | 理論値: 23.312551 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: AQSPATISLP QGGQFRLSIS NTDPNMIFIP GDKVTAITAP GGMLADKRLT TAGGVLFTSV ATRTFTIFVE TALGQTFSVV ATPVKGEGR VYRLMSAEPP SRPETRKWET AQAYEKLLIS LNRAVLTGDI PDGYGEVKPL SDGIRLPGGF SVTPLKAWAG D QLRADRYE ...文字列: AQSPATISLP QGGQFRLSIS NTDPNMIFIP GDKVTAITAP GGMLADKRLT TAGGVLFTSV ATRTFTIFVE TALGQTFSVV ATPVKGEGR VYRLMSAEPP SRPETRKWET AQAYEKLLIS LNRAVLTGDI PDGYGEVKPL SDGIRLPGGF SVTPLKAWAG D QLRADRYE LRNANTWGVA LREQDFWKPG VRAVMFDNNA QTLMGGGRMT VTVIRGNG UniProtKB: Type-F conjugative transfer system secretin TraK |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 1.3 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: SPOT SCAN / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)